We all know that CO2 dumped in the atmosphere (consequences in the ocean, where the most carbon winds up actually are even dire – i.e. ocean acidification) causes global warming through what’s commonly referred to as the greenhouse gas effect. Governments and various environmental panels have through out the years issued various policies meant on curbing emissions. Ironically, however, greenhouse gas emissions have only gone up, as year after year there seems to be a new record in how much CO2 gets released into the atmosphere, mainly due to developing countries catching up and becoming industrialized. Only recently, the world passed a frightening threshold after atmospheric CO2 levels reached 400ppm (parts per million) for the first time in 3 million years.
The reason I’m presenting these facts isn’t to inflict panic. Indeed, these are depressing data, however it’s important to build context especially when covering cutting edge research conducted by scientists working effortlessly to battle atmospheric CO2 dumping. One of the most creative solution is the development of carbon trapping technology, and exciting as the tech may be it still bears a grand challenge: what to do with the stored carbon. While more efficient plants and filters significantly cut down emissions, you still windup with excess carbon – sure it’s not in gaseous form as a CO2 compound, so it doesn’t contribute to the greenhouse gas effect, but you still need to get rid of it.
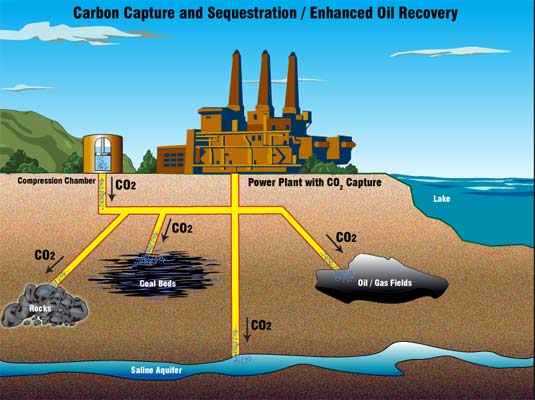
Some byproducts get pumped into the ground directly, where it seeps though cracks of rock layers deep below the surface, a process that we already know causes huge chemical changes in the rock. Other methods involve dumping the stored carbon by pumping it into the ocean, where pressures below a certain depth will cause it to form a thick slurry that falls to coat the ocean floor – in theory, right at the bottom, it’s harmless. The method is promising when you need to dump a few tons of carbon, but at the massive industrial scale you need a system capable of disposing millions of tones of carbon – hint: it needs to be cheap; dirt cheap!
Researchers at University of Newcastle have come up with a new solution that not only elegantly solves this problem, but offers a practical use. What if instead of dumping the captured carbon you turn it into something useful? This is exactly the reasoning behind the Newcastle researchers project, recently awarded a $9 million grant, inspired by nature, namely the sequestration of CO2 as rocks of neutral carbonate in the Earth itself.
Using CO2 to construct the buildings of the future
Reacting gaseous CO2 with low grade minerals such as magnesium and calcium silicate produces limestone. The scientists’ idea is to exploit the process by combining their captured carbon with cheap minerals and voila! You’ve got some limestone-like material right at your disposal that you can fashion into bricks for construction purposes. You could use it for anything from buildings to paving. Limestone’s pretty resilient and strong, too. Actually, it has been used in everything from the Egyptian Pyramids to the British Parliament buildings.
A multidisciplinary research team, including Professors Bodgan Dlugogorski and Eric Kennedy from the University’s Priority Research Centre for Energy and Orica Senior Research Associate Dr Geoff Brent, have demonstrated the technology in small scale laboratory settings and led the funding bids.
“The key difference between geosequestration and ocean storage and our mineral carbonation model is we permanently transform CO2 into a usable product, not simply store it underground,” Professor Dlugogorski said.
Brilliant, but how come anyone didn’t think of this before? They have, but the problem has always been that the process is highly energy intensive. This means its expensive since you need to produce a lot of energy to funnel the process, then producing this energy typically implies burning fossil fuels, which releases carbon. You can understand how unreasonable the idea becomes. The breakthrough though is that the Newcastle researchers have devised a way of dramatically lowering the energy threshold. This same year, Newcastle made the production of calcium carbonates “a thousand times cheaper” through the use of nickel nanoparticles. A similar process is employed for the “carbon limestone”.
“The Earth’s natural mineral carbonation system is very slow,” Professor Kennedy said. “Our challenge is to speed up that process to prevent CO2 emissions accumulating in the air in a cost-effective way.”
It might take a while before this process might catch on, however. For now, Newcastle plans on using its grant to built a mineral carbonation research pilot plant, expected to open in 2017. There the researchers can extend the findings in the lab to an environment similar to that found in the industry. If they can manage to produce mineral carbonation at a price that’s under current construction materials then they’ve hit the jackpot!
[READ ON] Carbon negative: removing CO2 altogether from the atmosphere






