New research from Caltech could bring an economically-viable solar fuel to the market in the next few years.

One of the holy grails of renewable energy researchers which they have been pursuing for decades, is the brewing of economically-viable solar fuel. It sounds like something you drill out of the core of a star, but in reality, it’s both much more useful and less dramatic than that: “solar fuels” are chemical compounds which can be used to store solar energy.
Most of the recent research performed in this field focused on splitting water into its constituent parts (hydrogen and oxygen) using only sunlight. It’s easy to see why — hydrogen produced this way would be a clean, cheap, easy-to-produce and generally widely-available fuel. It could be used to power solar cells, motor vehicles, or even spin the turbines of power plants. One of it’s most attractive qualities is that it would be virtually endless and produces zero emissions: the only reactionary product of a hydrogen engine (which burns the gas, i.e. combines it back with oxygen) would be plain old water.
We’re actually pretty close to having the solar fuels we so desire, the only thing we’re missing is the “cheap” part. Back in 2014, a team of Caltech researchers led by Professor Harry developed a water-splitting catalyst from layers of nickel and iron. It worked pretty well for a prototype, showing that it has potential and could be scaled-up. However, while the catalyst clearly worked, nobody knew exactly how it did so. The working theory was that the nickel layers were somehow responsible for the material’s water-splitting ability.
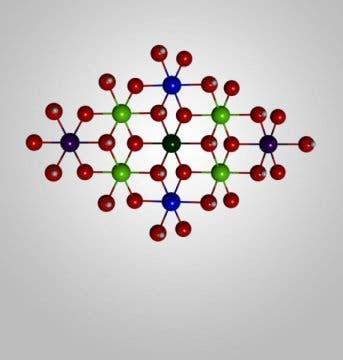
Image credits Caltech.
To get to the bottom of things, a team led by Bryan Hunter from Caltech’s Resnick Institute created an experiment during which the catalyst was starved of water, and observed how it behaved.
“When you take away some of the water, the reaction slows down, and you are able to take a picture of what’s happening during the reaction,” Bryan says.
The experiment revealed that the spot where water gets broken down on the catalyst — called its “active site” — wasn’t nickel, but iron atoms. The results are “very different” from what researchers expected to find, Hunter says. However, that isn’t a bad thing. Our initial hypothesis was a dud, but now that we know exactly how the alloy works — meaning we won’t waste time researching the wrong avenues.
“Now we can start making changes to this material to improve it.”
Gray believes the discovery will be a “game changer” in the field of solar fuels, alerting people that iron is “particularly good” for this type of applications. As we now know what we should look for, we can go on to the next step — which is finding out how to make such processes unfold faster and more efficient, which translates to lower costs of the final fuel.
“I wouldn’t be at all shocked if people start using these catalysts in commercial applications in four or five years.”
The paper “Trapping an Iron(VI) Water Splitting Intermediate in Nonaqueous Media” has been published in the journal Joule.






