An international team of researchers has uncovered the reactionary processes biology uses to bind gold. The process, which is employed by heavy-metal-resistant bacteria, could revolutionize how we extract the precious metal.

Most organisms don’t really like high concentrations of heavy metals such as copper or gold, as they are quite toxic and pose a significant threat. However, certain beings, such as the bacterium C. metallidurans, have adapted to withstand very high concentrations with ease. It does this by extracting useful elements from compounds laden with heavy metals and then depositing the toxic bits.
One interesting (and potentially quite lucrative) side-effect of this process is that C. metallidurans excretes pure gold, which coalesces into nuggets. Knowing how fond humans are of shiny things, a team of researchers from the Martin Luther University in Halle-Wittenberg (MLU), the Technical University of Munich (TUM), and the University of Adelaide in Australia have identified the molecular processes that underpin this nuggeting.
The bug with the golden eggs
C. metallidurans is a rod-shaped bacterium that primarily colonizes soils enriched/polluted with numerous heavy metal compounds. While that sounds like the bacterium is a poor judge of real-estate, its choice in soil actually comes with significant perks. Chief among them is that other bacteria don’t want anything to do with this harsh environment. The second is that if you look past the “deadly toxic” aspect, heavy metal compounds are a surprisingly hearty source of energy.
“Apart from the toxic heavy metals, living conditions in these soils are not bad,” explains Professor Dietrich H. Nies, a microbiologist at MLU. “There is enough hydrogen to conserve energy and nearly no competition. If an organism chooses to survive here, it has to find a way to protect itself from these toxic substances.”
It was actually Nies himself, together with co-author Frank Reith, a Professor at the University of Adelaide, who found that C. metallidurans can deposit gold biologically back in 2009. However, they were unable to say why or how it did so — something they addressed in their new paper.
The process actually starts with copper. Copper is a vital trace element for C. metallidurans, however, the form it’s usually found geologically can’t be easily absorbed and processed by the bacteria. So the bacteria unleashes a barrage of chemical processes meant to soften up this copper and convert it to a form that’s easily gobbled up.
Gold enters the bacteria using the same processes and membrane channels as copper, the team reports. These compounds, by and large, resemble the copper ones, and so are processed by the same chemicals the bacterium uses for copper. Also, just like copper, gold becomes very toxic very fast at higher concentrations. The only difference is that C. metallidurans don’t actually want or need gold.
To make sure there isn’t too much of a copper buildup inside its membrane, the bacteria use an enzyme called CupA to pump any excess out, the team reports. However, when both metals are present in the cell, the CupA enzyme becomes suppressed, Nies explains. This poses quite a problem for the bacteria as the two metals “combined are actually more toxic than when they appear on their own.”
To flush out this extra-toxic cocktail, C. metallidurans employs a second enzyme dubbed CopA. This transforms both metals back into their original, hard-to-absorb forms.
“This assures that fewer copper and gold compounds enter the cellular interior,” Nies explains. “The bacterium is poisoned less and the enzyme that pumps out the copper can dispose of the excess copper unimpeded.”
“Another consequence: the gold compounds that are difficult to absorb transform in the outer area of the cell into harmless gold nuggets only a few nanometres in size.”
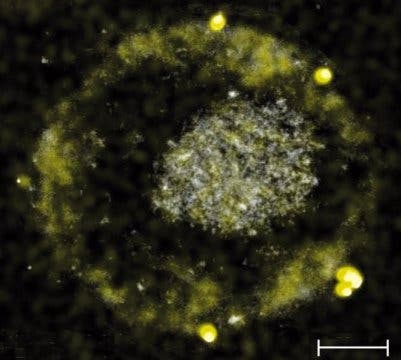
Image credits L. Bütof et al., 2018, Metallomics.
The whole process takes toxic gold particles formed by erosion or other weathering processes and turns them into harmless gold nuggets. These nuggets are considered to be secondary gold, so called because they’re generated from primary (geologically-created) gold, broken down from ores and re-deposited.
The research completes our understanding of the biogeochemical gold cycle — the second half of which was largely unknown up to now. This cycle sees a bacterial transformation of primary gold metal into toxic compounds, followed by another, bacteria-powered transformation into secondary metallic gold in the last half of the cycle.
Fully understanding the processes involved in this cycle could help revolutionize the way we extract gold, making the process cleaner and more efficient. The most significant advantages over today’s methods would be the possibility to use poorer ores (with a small percentage of gold) than before and taking mercury and other very toxic compounds out of the extraction process.
The paper “Synergistic gold–copper detoxification at the core of gold biomineralisation in Cupriavidus metallidurans” has been published in the journal Metallomics.






