After they identified precise groups of cells in mice brain that induce eating and others that curb it, a team of researchers caused full mice to continue eating and hungry mice to stop eating simply by stimulating one of these areas. Their findings could aid in the development of novel drugs that target eating disorders such as anorexia or obesity.
“This is a really important missing piece of the puzzle,” says neuroscientist Seth Blackshaw of Johns Hopkins University in Baltimore. “These are cell types that weren’t even predicted to exist.”
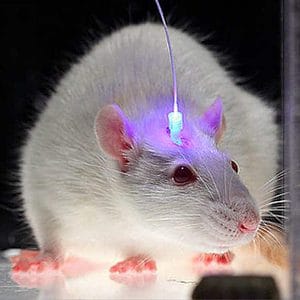
Scientists led by Joshua Jennings and Garret Stuber of the University of North Carolina at Chapel Hill first genetically engineered mice such that some of their neurons responded to light. This allowed them to apply optogenetics techniques to manipulate the behavior of these neurons. Optogenetics is a hot technique in neuroscience research right now, involving taking a light-activited gene (called a channel rhodopsin) targeted into a single neuron type, and inserting it into the genome of, say, a mouse. Whenever a light is shone into the mouses’s brain, the rhodopsin channel replies and consequently the neurons expressing the rhodopsin channel fire. Basically, simply by firing light you get certain neuron types to become activated or not (there are channels that inhibit neuron firing).
An on/off switch for eating
The neurons the researchers targeted reside in a brain locale called the bed nucleus of the stria terminalis, or BNST. Some of the message-sending arms of these neurons reach into the lateral hypothalamus, a brain region known to play a big role in feeding. When a laser was used to activate BNST, the mice began to insatiably eat.
“As soon as you turn it on, they start eating and they don’t stop until you turn it off,” Stuber says.
The opposite behavior happened when a laser silenced BNST neurons’ messages to the lateral hypothalamus: The mice would not eat, even when hungry. These two important observations back-up the idea that these lateral hypothalamus neurons normally restrict feeding.
Is there a physiological feeding limit though? For instance, if these neurons were stimulate indefinitely would the mice die from overeating or starvation? The researchers don’t know since for this particular research they only used short 20-minute-long bursts of laser light. Longer-term manipulations of these neural connections — perhaps using a drug — might cause lasting changes in appetite and, as a result, body mass, Stuber says.
What the findings, which were reported in a paper published in the journal Science, once again show is the brain is the master. We think of feeding in terms of metabolism and body stuff,” Stuber says. “But at the end of the day, it’s controlled by the brain.”
via Science News






