The shell of a virus which calls boiling acid pools of Yellowstone home could show us the way to more powerful drugs, stronger materials, and more resilient buildings.
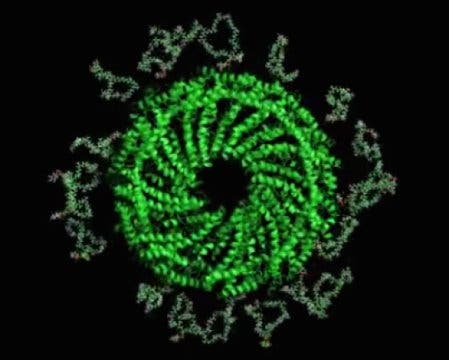
Image credits Peter Kasson et al., 2017.
First isolated in 2002 by Pasteur Institute researchers, the virus known as Acidianus hospitalis Filamentous Virus 1 is scaringly hard to kill. This bug lives in the hot-springs of Yellowstone National Park, pools of acid bubbling to temperatures as high as 237 degrees Celsius (455-ish degrees Fahrenheit), which can dissolve people.
Naturally, anything that can live in such conditions is bound to have some pretty impressive tricks up their sleeves — or up their viral casing, as an international team of researchers reports. The finding could open up new avenues of research into material sciences, with applications from pinpoint drug delivery mechanisms to earthquake-resistant buildings.
“Anytime you find something that behaves really differently, especially something this stable, it’s interesting and potentially useful,” says first author Peter M. Kasson, MD, PhD, from the University of Virginia School of Medicine Department of Molecular Physiology and Biological Physics
“When you’re doing curiosity-driven science that finds something new, in the back of your mind, you think, ‘Hey, this is really different. What might it be good for?’ And this has many potential applications.”
Thick-skinned
The secret behind the virus’ extreme resilience lies in its membrane, the team reports. Although it’s outer shell is only half as thick as known cell membranes, it’s ridiculously stable. The molecules which make up the membrane are stacked up in a horseshoe-shaped arrangement, creating a very dense and durable structure.
The team had to rely on the UVA’s Titan Krios electron microscope to probe the virus’ secrets. This device is so sensitive, that it had to be installed underground to insulate it from vibrations — the slightest of which would be enough to throw off its calibrations. Armed with these readings, the team turned to computer modeling to tease out the structure of the membrane’s lipid molecules.
“Essentially, we encode everything we know about the physics of these molecules and then come up with models that are both consistent with the basic physics and consistent with the observations from the electron microscope,” Kasson explains.
Duplicating this structure might help scientists paste the virus’ defenses into other materials. These could have a dramatic impact in construction, material science, every field where a super-resilient material could come in handy.
Nanomedicine stands to benefit a whole lot if we manage to reverse-engineer the virus’ shell. We could use the structures to create microscopic particles to protect drugs from our body’s effort to metabolize them, allowing for pinpoint delivery of more efficient drug doses. For example, injecting drugs directly into tumors.
“It’s amazing how much we still don’t know about life as it exists on Earth — at the bottom of the ocean, in the deep sea vents, or places like Yellowstone or Iceland where you have these very strange environments we think of as inhospitable to life,” paper co-author Edward Egelman said.
“But the things that live there, they may look at our environment and think, ‘Strange.'”
The paper “Model for a novel membrane envelope in a filamentous hyperthermophilic virus” has been published in the journal eLife.






