Inspired by comic book heroes from the Marvel Universe, researchers at the University of California, Riverside, have developed a self-healing polymer that can stretch 50 times its normal size. The material could be used to create everyday electronics such as smartphones that repair themselves when dropped or compromised in some other way. Soft robotics, a blooming field of science, is also set to benefit from such a material.

“When I was young, my idol was Wolverine from the X-Men,” Chao Wang, Ph.D., says. “He could save the world, but only because he could heal himself. A self-healing material, when carved into two parts, can go back together like nothing has happened, just like our human skin. I’ve been researching making a self-healing lithium ion battery, so when you drop your cell phone, it could fix itself and last much longer.”
Our bodies are excellent biological machines that need little (conscious) maintenance. That’s because they heal any injury by themselves and have self-repair mechanisms set in place to kill rogue cells, fight infectious agents, repair broken proteins, keep our coronary arteries open and naturally fight the aging process — day in, day out. A broken piece of plastic, however, doesn’t have an armada of eager cells at its disposal to mend it back to one piece. But there are chemical means that can make the plastic whole again.
The key to self-repair has to do with how atoms and molecules bond together. Loosly speaking, there are two types of chemical bonds, namely covalent and non-covalent. Covalent bonds, the most common type of bond in organic molecules, involves the sharing of electrons between two atoms. They’re strong, but because of their rigid nature covalent bonds don’t readily reform once they’ve been broken. Non-covalent bonds, on the other hand, are weaker but more dynamic.
Water molecules, for instance, are tied together through hydrogen bonds — which are non-covalent. This type of bond breaks and reforms constantly, which is why water has such fluid properties.
With this in mind, many researchers have made attempts at self-healing polymers that form hydrogen bonds, but these are not suitable as ionic conductors, which is something you want in an electronic device.
Instead, Wang and colleagues turned to a different type of non-covalent bond called an ion-dipole interaction, a force that binds charged ion to polar molecules. These interactions have never been exploited for designing self-healing polymers, but Wang says these turn out to be very suitable for ionic conductors. The breakthrough lied in using a polar, stretchable polymer, poly(vinylidene fluoride-co-hexafluoropropylene), plus a mobile, ionic salt. The polar groups in the polymer and the ionic salts are linked together by the ion-dipole interactions.
Tests suggest the resulting material can stretch to 50 times its original size. When torn in two, the material stitches itself back together in its original configuration within a day. No superglue required.
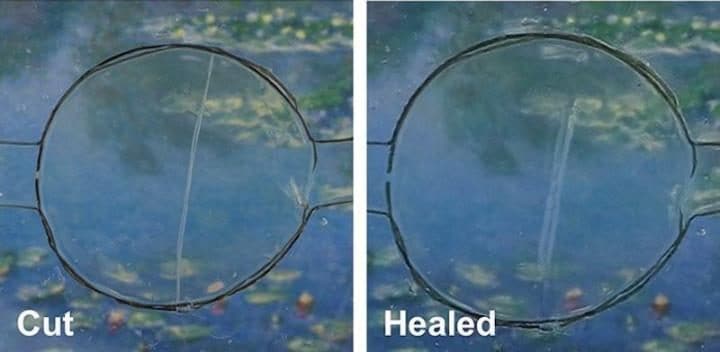
The behavior is analogous to how human skin heals. If the material were to be used in a smartphone, it could fuse scratches by itself — a very common problem for many smartphone users, who usually end up replacing their devices because of it. Wang and colleagues are actually working on a self-repairing lithium-ion battery, another common component which gets damaged often.
To test the material in a more practical setting, the researchers placed a non-conductive membrane between two layers of the ionic conductors. When current was applied, the sandwiched bulk material responded with movement. Effectively, they had made a so-called artificial muscle.
Next on the drawing board is altering the polymer to improve its properties. The researchers will also test it under harsher conditions such as high humidity — which has renderered previous self-healing polymers useless.
“Water gets in there and messes things up. It can change the mechanical properties. We are currently tweaking the covalent bonds within the polymer itself to get these materials ready for real-world applications,” Wang says.
The authors described their findings at the 53rd National Meeting & Exposition of the American Chemical Society (ACS).






