A new paper describes how one species of bacteria learned to not only stay safe but profit from their greatest enemies, amoeba. The secret is wielding a lot, a lot of knives.

Free to use with attribution.
Bacteria usually steer as clear of amoeba as they possibly can — because amoeba like to snack on them. They’re bigger and usually faster than bacteria, and will envelop any they can catch in pseudopodia to be digested at a later date. Not the nicest of fates.
Some of the bacteria have learned to defend themselves in pretty spectacular ways. One such species is known as Amoebophilus, and was discovered by researchers at the University of Vienna a few years back. It doesn’t only survive inside amoeba but is so well adapted to thrive in there that they’ve have become this species’ favorite habitat.
Now, working together with the Viennese researchers that discovered the bacterium, scientists from ETH Zurich have uncovered the mechanism that guarantees the bug’s survival inside amoeba. And it would make Arya Stark proud: the bacteria has (possibly poison-coated) dagger-launcher devices built into its membrane that allows it to shred their predators from inside and avoid digestion.
Bringing a knife to a bug fight
To find the bacteria’s secret, the researchers used a novel method developed at ETH that’s currently in use only in a handful of labs worldwide. They froze amoeba that had absorbed bacteria at minus 180°C (-292°F). Then João Medeiros, a doctoral student at ETH and paper co-author, used a focused ion beam to cut away at the specimens, much like archaeologists chip away at soil and rock around an artifact with chisels. After milling away the amoeba and most of the bacteria, Medeiros was able to extract the bug’s defensive mechanism and produce a three-dimensional electron tomogram of it.
The mechanism consists of several sheaths affixed onto the bacterium’s inner membrane by a baseplate and an anchoring platform. Their purpose is to shoot molecular-sized “daggers” at the amoeba. Each sheath “is spring-loaded and the micro-dagger lies inside it,” explains João Medeiros, a doctoral student at ETH and paper co-author.
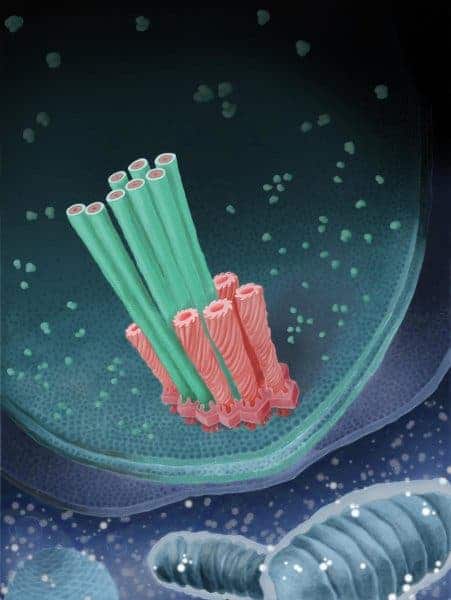
Image credits Leo Popovich / ETHZ.
“When the sheath contracts, the dagger is shot outwards extremely quickly through the bacterial membrane.”
This comes in really handy for the tiny Amoebophilus, as amoeba surround their pray into a stomach-like digestive vesicle (a pocket of membrane inside the amoeba filled with digestive fluids). The daggers shred through these vesicles and allow the bacteria to escape the very unfriendly conditions of the digestive bubble. But it doesn’t escape all the way — once safe, Amoebophilus stays inside the amoeba and takes advantage of its host to feed and even multiply.
We don’t really know why these digestive vesicles get destroyed so easily. It may be that it’s simple mechanical force that does it, but the team suspects there’s more at play here. Their theory is that Amoebophilus’ daggers are also coated in ‘poison’ in the shape of membrane-degrading enzymes. The theory looks to be right, based on bits of genetic code that researcher at the UOV have shown to encode information for such enzymes.
Tiny stabbings
It’s not the first time we’ve seen such daggers in biology. Bacteriophages, viruses which specialize in preying on bacteria, use similar systems to inject their genome past their defenses. Some species of bacteria are known to use similar micro-weapons to pepper their surroundings in ‘ow’ and deter competing microorganisms from a source of food, for example.
What sets these findings apart is the sheer fidelity and resolution the team managed to get on the mechanisms. They’re also the first to create the complete 3D structure of such mechanisms inside cells in their natural context and the first to show the details in the baseplate and membrane anchor.
“In the past, cell biologists investigated the function of such systems and structural biologists elucidated the structure of individual components,” says ETH Professor Martin Pilhofer, the paper’s corresponding author.
“With the cryo-focused ion beam milling and electron cryo-tomography technologies that we have established at ETH Zurich, we can now close the gap between cell biology and structural biology.”
Another new discovery the team can boast is finding these mechanisms arranged in clusters. All previous known micro-dagger weaponry we’ve found is deployed as individual devices. Amoebophilus, however, is not shy of escalating the conflict, using heavy batteries or clusters of up to 30 dagger-launching systems.
“You could call them multi-barrel guns,” Pilhofer adds.
The team also used genomic comparisons to peek into how Amoebophilus evolved its daggers. Their results show that “the relevant genes are very similar to bacteriophage injection systems,” which led the team to believe that these genes are originated from an ancestor of today’s bacteriophages that left bits of its code in the bacteria’s genome long ago. The results also suggest that many other bacterial strains in at least nine of the most important groups should have similar dagger arsenals. It’s not yet known whether they serve the same purpose or not, but the team says their next steps will be to find out.
They also plan to use the new method of cryo-focused ion beam milling to look at other complex molecular systems.
The full paper “In situ architecture, function, and evolution of a contractile injection system” has been published in the journal Science.






