Antibiotics are medicines that combat infections caused by bacteria. However, due to misuse and overuse of antibiotics, many bacterial strains are developing antibiotic resistance.

Before Alexander Fleming discovered penicillin in 1928, there was no effective treatment for infections such as pneumonia, gonorrhea or rheumatic fever. Fleming’s discovery kicked off a golden age of antimicrobial research with many pharmaceutical companies developing new drugs that would save countless lives. Some doctors in the 1940s would famously prophesize that antibiotics would finally eradicate the infectious diseases that had plagued humankind throughout history. Almost a hundred years later since Fleming made his milestone discovery not only are bacterial infections still common, the misuse and overuse of antibiotics are threatening to undo all of this medical progress as bacterial strains become resistant.
Antibiotic resistance: a modern problem that can be traced to ancient times.
Contrary to common belief, human exposure to antibiotics isn’t confined to the modern era. Traces of tetracycline, a broad-spectrum antibiotic, have been found in the skeleton remains from ancient Sudanese Nubia dating from 350-550 CE. Likewise, tetracycline has been found in remains dating from the late Roman period in the Dakhleh Oasis, Egypt. These people must have included tetracycline in their diet — and it was to their good fortune as the rate of infectious diseases documented in Sudanese Nubian populations was low. For thousands of years, Chinese herbalists have been using a variety of plants which contain antimicrobial active components for ancient traditional remedies.
Naturally, the selective pressure imposed by these ancient antimicrobial activities has led to the accumulation of antibiotic resistance genes. But that’s nothing like the scale and intensity of antibiotic resistance we’re seeing today.
What is antibiotic resistance
Antibiotic resistance occurs when an antibiotic is no longer effective at controlling or killing bacterial growth. Bacteria which are ‘resistant’ can multiply in the presence of various therapeutic levels of an antibiotic. Sometimes, increasing the dose of an antibiotic can help tackle a more severe infection but in some instances — and these are becoming more and more frequent — no dose seems to control the bacterial growth. Each year, 25,000 patients from the EU and 63,000 patients from the USA die because of hospital-acquired bacterial infections which are resistant to multidrug-action. The ECDC/EMA Joint Working Group estimated in 2009 that the cost due to multidrug-resistant bacterial infections amounts to EUR 1.5 million in the EU alone. According to a 2013 CDC report titled “Antibiotic Resistance Threats in the United States“, antibiotic resistance is responsible for $20 billion in direct health-care costs in the United States.
Antimicrobial resistance threatens to undermine all the immense clinical and public health progress we’ve come to achieve so far. This is a very complex problem that requires concentrated and coordinated efforts of microbiologists, ecologists, health care specialists, educationalists, policy makers, legislative bodies, agricultural and pharmaceutical industry workers, and the public to deal with.
The main challenges in dealing with antibiotic resistance are, on one hand, genetically acquired immunity and, on the other hand, fewer and fewer novel drugs. Since the 1970s, the rate at which new antibiotic classes have been discovered has continued to drop. No novel drug classes have been developed in the last 20 years. Researchers nowadays agree that, at this current rate, humanity is destined to lose the arms race as sooner or later bacteria will acquire resistance to modified versions of currently available antibiotic classes.
How bacteria develop resistance to antibiotics
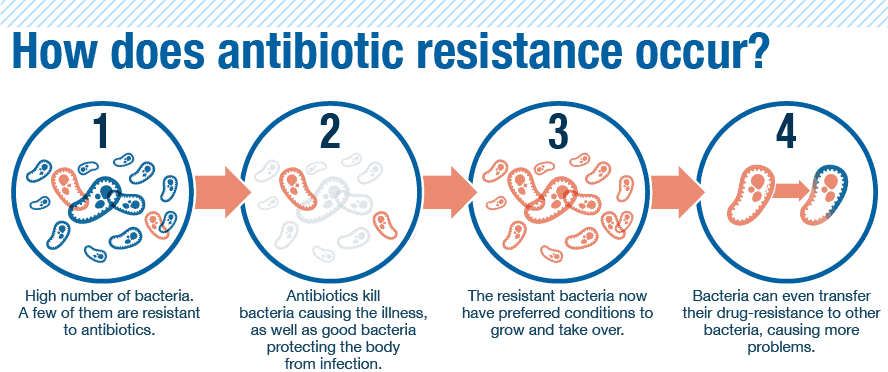
Every time a person takes antibiotics, sensitive bacteria are killed, but resistant germs may be left to grow and multiply. In time, these leftover populations can become so strong that antibiotics no longer are effective.
There are several mechanisms bacteria employ to become resistant. Some gain the ability to neutralize the drug before it gets the chance to attack the bacteria. Other bacteria can rapidly pump the antibiotic out or can even change the attack site so the function of the bacteria isn’t affected.
Whenever bacteria survives an antibiotic onslaught, it can acquire resistant through mutation of the genetic material or by ‘borrowing’ pieces of DNA that code for the resistance to antibiotics from other bacteria, like those from livestock. Moreover, the DNA that codes the resistance is grouped in an easily transferable package which enables the germs to become resistant to many antimicrobial agents.
The types of bacterial resistance
- Intrinsic resistance. Some bacteria are intriguingly resistant to antibiotics, such as those that don’t build a cell wall (penicillin prevents cell-wall building).
- Acquired resistance. Bacteria can acquire resistance through new genetic change or by transferring DNA from a bacterium that is already resistant. This is the issue we’re having today.
According to the CDC, the following bacterial strains have developed the most resistance such that they’ve been listed as urgent hazards:
- Clostridium difficile. Causes severe diarrhea, especially in older people and those who have serious illnesses.
- Enterobacteriaceae. These normally live in the digestive tract but can invade other parts of the body, like the urinary tract, and cause infections.
- Neisseria gonorrhoeae. Causes gonorrhea, a sexually transmitted infection. In 2016, the WHO said gonorrhea might soon become untreatable.
Why antibiotic resistance is growing
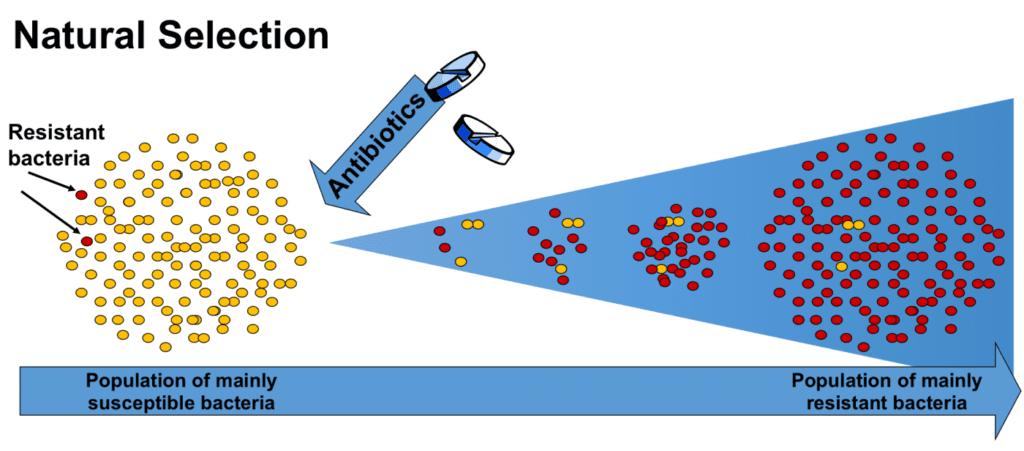
There are a number of factors that contribute to this growing health hazard. Among them we can mention:
- self-medication;
- hygienic habits such as the use of anti-bacterial soap which research suggests is useless but significantly contributes to the growing problem of antimicrobial resistance;
- counterfeit drugs, particularly rampant in the developing world;
- antibiotics for livestock;
- infections acquired in hospitals and nursing homes, particularly in the developed world;
There’s no surprise in the fact that antibiotic resistance infections correlate with the level of antibiotic consumption. The more antibiotics a population consumes, the faster bacteria will adapt and become resistant. One huge problem is the mindless use of antibiotics. For instance, many patients request their doctors to prescribe antibiotics when there is no need for them, such as in the case of viral infections. Research shows that up to 15 million people in the United States go to the doctor for a sore throat every year. About 70 percent of these patients receive strep throat antibiotics but only 20 percent actually have strep throat, according to the IDSA.
Another problem is compliance with strict drug regimes. To be effective, antibiotics needs to be taken at least over several days and the scheduling needs to be respected on the clock yet many patients fail to follow these instructions.
Things are worse in some countries than others. For instance, in some countries, antibiotics are available without a prescription so the potential for self-medication abuse is huge especially if the patient is not educated about antibiotics. In the absence of a proper diagnosis, suitable antibiotic choice, correct usage, compliance, and treatment efficiency monitoring, self-medicating antibiotics can only exacerbate the mounting resistance problem.
Another issue lies with antibiotics for domestic animals, particularly livestock. Farmers widely use antibiotics to stave off infections but also for promoting growth. Approximately 80 percent of the antibiotics sold in the United States are used in meat and poultry production, and in the vast majority of cases, the antibiotics are used on healthy animals. This practice can lead to the evolution of ‘superbugs’ which can migrate into the environment as people consume meat.
In 2003, an Expert Workshop co-sponsored by the World Health Organization, Food and Agricultural Organization (FDA), and World Animal Health Organization (OIE) concluded “that there is clear evidence of adverse human health consequences due to resistant organisms resulting from non-human usage of antimicrobials. These consequences include infections that would not have otherwise occurred, increased frequency of treatment failures (in some cases death) and increased severity of infections”
Most recently in 2012, the FDA stated “Misuse and overuse of antimicrobial drugs creates selective evolutionary pressure that enables antimicrobial resistant bacteria to increase in numbers more rapidly than antimicrobial susceptible bacteria and thus increases the opportunity for individuals to become infected by resistant bacteria.”
Solutions to antibiotic resistance
The sad reality today is that there’s not much we can do for patients who don’t respond to antibiotics, which is why mortality rates are so high.
“Antibiotic resistance is rising for many different pathogens that are threats to health,” said CDC Director Tom Frieden, M.D., in a statement. “If we don’t act now, our medicine cabinet will be empty and we won’t have the antibiotics we need to save lives.”
Some researchers have proposed alternatives to antibiotic treatment such as passive immunization or phage therapy but most efforts are directed towards the discovery of new and more efficient antibiotics. Like outlined earlier, however, most of our antibiotics have been isolated in the so-called ‘golden era’ of antibiotic discovery from a limited number of taxonomic groups, mainly from Actinomyces that live in the soil. Some research groups are exploring alternative ecological niches such as the marine environment. Other approaches involve borrowing antimicrobial peptides and compounds from animals and plants, as well as the natural lipopeptides of bacteria and fungi. There is also a potential to find new antibiotics by exploring the microbiota through the metagenomic approach. Finally, some groups are looking design new classes of antibiotics from scratch through complete synthesis.
Preventing antibiotic resistance
Finding new antibiotics, however, will likely not solve our growing antibiotic resistance problem. History has shown that after a new antibiotic therapy is introduced, sooner or later resistance will arise. This approach is destined to fail since bacteria will eventually respond to selective pressure by the emergence of resistance mechanisms.
What we can do, however, is to buy time until someone very clever figures a way to outsmart bacteria for good.
Scandinavian countries, for instance, banned the use of growth-promoting antibiotics in livestock since 2006 and other EU countries have been implementing similar measures. In 2012, the FDA ruled that certain extra-label uses of cephalosporin antimicrobial drugs should be banned from certain livestock.
It is estimated that in half of all cases, antibiotics are prescribed for conditions caused by viruses. Obviously, in such cases the antibiotics are useless and doctors and nurses ought to know better.
Governments have a critical role in combating antibiotic resistance. It’s imperative that robust action is taken both at a national and international level in order to regulate the appropriate use of quality medicines and education about the dangers of overuse. A lot of antibiotic resistance is building up in developing countries where there is little oversight. Governments need to work together to strengthen the health care quality in such places for the good of us all. Not least, the industry needs to move faster and more aggressively to research and develop new antibiotics.
What you can do
- Don’t take antibiotics for a viral infection like a cold or the flu.
- Do not save any antibiotics for the next time you get sick. Discard any leftover medication once you have completed your prescribed course of treatment
- Always take antibiotics only after you’ve consulted with a health care professional. The FDA has a great campaign called “Get Smart: Know When Antibiotics Work” that offers Web pages, brochures, fact sheets, and other information sources aimed at helping the public learn about preventing antibiotic-resistant infections.
- Take an antibiotic exactly as the healthcare provider tells you. Do not skip doses.
- Never pressure your provider to prescribe an antibiotic.
- Never use antibacterial soap.






