Researchers at the University of Bristol used a complex computer simulation to determine how enzymes in bacteria breakdown antibiotics, rendering them useless. The same simulation could be used to see how certain drugs respond to various bacteria, predicting their resistance and efficacy. This way, doctors will be able to prescribe the best antibiotics for a particular outbreak, saving time, energy, money and lives.
How bacteria react to antibiotics
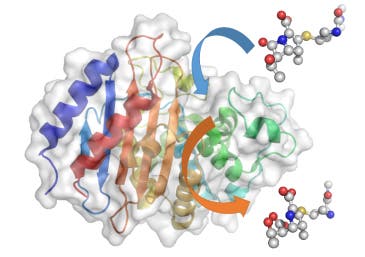
The team used a Nobel Prize-winning technique called QM/MM – quantum mechanics/molecular mechanics simulations – to gain a molecular-level insight into how enzymes called ‘beta-lactamases’ react to antibiotics. Resistance to antibiotics is an ever worrisome topic among medical experts since it can lead to a new pandemic nightmare. There are numerous infections that are nowadays commonly treated by antibiotics, and since Alexander Fleming’s breakthrough discovery of penicillin millions of lives have been saved. Because too many people take antibiotics when they don’t actually need them or because the wrong antibiotics are prescribed, studies report a growing trend that suggests bacteria are becoming ever adaptive. As a line of ‘last-resort’ physicians often prescribe a class of antibiotics called carbapenems for cases where any other antibiotic doesn’t respond- this was the focus of the researchers at University of Bristol.
The QM/MM simulations revealed that the most important step in the whole process is when the enzyme ‘spits out’ the broken down antibiotic. If this happens quickly, then the enzyme is able to go on chewing up antibiotics and the bacterium is resistant. If it happens slowly, then the enzyme gets ‘clogged up’ and can’t break down any more antibiotics, so the bacterium is more likely to die. The rate of this ‘spitting out’ depends on the height of the energy barrier for the reaction – if the barrier is high, it happens slowly; if it’s low, it happens much more quickly, according to the paper published in Chemical Communications.
Professor Adrian Mulholland, from Bristol University’s School of Chemistry, said: “We’ve shown that we can use computer simulations to identify which enzymes break down and spit out carbapenems quickly and those that do it only slowly.
“This means that these simulations can be used in future to test enzymes and predict and understand resistance. We hope that this will identify how they act against different drugs – a useful tool in developing new antibiotics and helping to choose which drugs might be best for treating a particular outbreak.”
Was this helpful?



