A new method of producing hydrogen has been reported by researchers at University of Glasgow that’s 30 times faster than current state-of-the-art methods, providing yet another advance that might one day lead to a sustainable hydrogen based economy.
There’s only so much that renewable energy can grow with today’s infrastructure due to base load considerations. If the intermittency can be compensated with a storage medium, then solar and wind power can be extended in safely plugged into the grid. Hydrogen is fantastic for energy storage and these latest developments definitely help us reach a point where the hydrogen economy is feasible and make renewable energy the prime mean of generating energy.
[ALSO READ] Researchers split water using device that runs on AAA battery
Hydrogen economy: pipe dream or way of the future?
The cleanest way to produce hydrogen is through electrolysis – an electrochemical method which uses electricity to break the bonds between water’s constituent elements, hydrogen and oxygen, and releases them as gas. There aren’t any environmentally harmful byproducts and the hydrogen can then be stored for later use in fuel cells, for instance, which work like electrolysis in reverse.
[RELATED] US navy synthesizes jet fuel solely from seawater
At an industrial level, the most popular method for generating hydrogen out of renewable power is by using proton exchange membrane electrolysers (PEMEs). These devices, however, require precious metal catalysts (platinum, palladium), and need to be subjected to high pressure and high densities of current. The new method allows larger-than-ever quantities of hydrogen to be produced at atmospheric pressure using lower power loads, typical of those generated by renewable power sources.
“The process uses a liquid that allows the hydrogen to be locked up in a liquid-based inorganic fuel. By using a liquid sponge known as a redox mediator that can soak up electrons and acid we’ve been able to create a system where hydrogen can be produced in a separate chamber without any additional energy input after the electrolysis of water takes place,” according to Professor Lee Cronin of the University of Glasgow’s School of Chemistry.
“The link between the rate of water oxidation and hydrogen production has been overcome, allowing hydrogen to be released from the water 30 times faster than the leading PEME process on a per-milligram-of-catalyst basis,” he added.
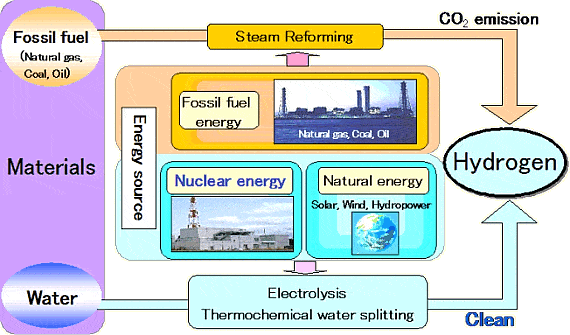
There’s a great demand for hydrogen in the world right now, especially for agriculture where the gas is an indispensable component of ammonia fertilizer production. Essentially hydrogen helps feed half the world, at least. But it could also power it. The main problem right now with hydrogen is that it’s far from being sustainable – the way it’s being produced that is. More than 95% of the hydrogen available today is made via steam-methane reforming, a mature production process in which high-temperature steam (700°C–1,000°C) is used to produce hydrogen from a methane source, such as natural gas. In steam-methane reforming, methane reacts with steam under 3–25 bar pressure (1 bar = 14.5 psi) in the presence of a catalyst to produce hydrogen, carbon monoxide, and a relatively small amount of carbon dioxide. So, what we’re seeing is a fossil fuel being subjected to a highly energy intensive treatment, whose energy most likely comes from fossil fuel combustion in the first place.
Developments such as these can only makes us happy and hope for a time where electrolysis systems can produce hydrogen cheaply and fast. Findings appeared in the journal Science.






