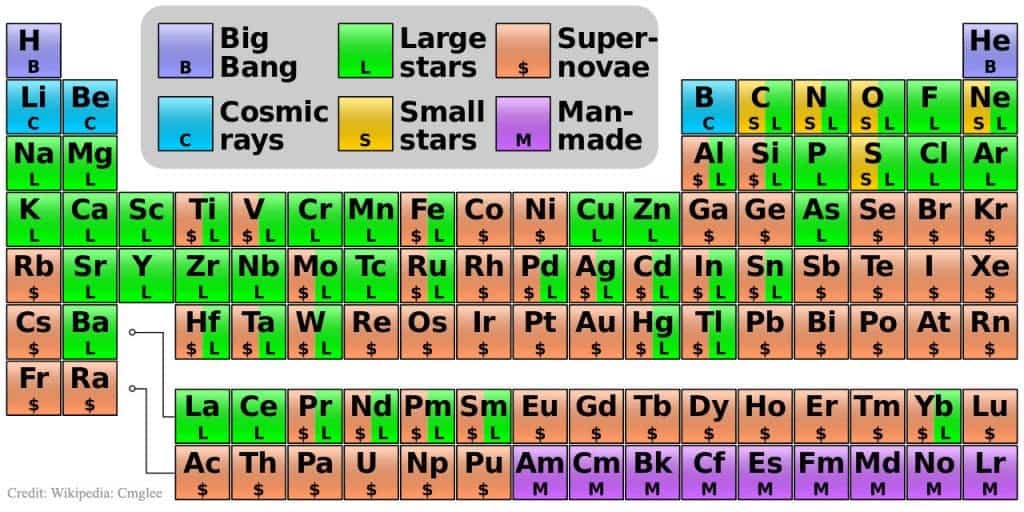
You may have heard about how we’re all made of star dust. That’s quite accurate, considering most elements that makeup the human body including carbon, oxygen or phosphorus were made by nuclear fusion inside the core of stars. Hydrogen, however, was made during the Big Bang, as well as helium along with traces of lithium and beryllium. Eventually, as the Universe cooled and expanded, cosmic dust and gases accreted and pressed by gravity formed stars.
For most of their time, stars are busying fusing hydrogen into helium. When they run out of hydrogen, stars begin to die. It will expand into a red giant star producing carbon atoms by fusing helium atoms. Those stars that are more massive have even more nuclear reactions and form elements ranging from oxygen to iron.
Nucleogenesis reactions
- 3 helium atoms fusing to give a carbon atom: 3 @ 4He → 12C
- carbon atom + helium atom fusing to give an oxygen atom: 12C + 4He → 16O
- oxygen atom + helium atom fusing to give a neon atom: 16O + 4He → 20Ne
- neon atom + helium atom fusing to give a magnesium atom: 20Ne + 4He → 24Mg
The last stellar evolutionary stages of a massive star’s life is marked with a boom: a supernova. As the star explodes it releases a massive amount of energy and neutrons, forming elements heavier than iron: uranium, gold etc.
[YOU SHOULD DEFINITELY CHECK OUT] 112 Elements from the Period Table Illustrated as Characters
So, 4 elements were made during the Big Bang, while 86 are made by stars. The rest of 28 out of 118 are thought to be man-made. That is, these have only been detected in labs in conditions staged by humans. Of course, these must form in nature as well, but only for a couple of moments before decaying into something else.
[via NASA’s APOD]