The hole in our planet’s ozone layer is getting patched up. At its peak this year, the hole grew to be two and a half times the size of the US territory, its smallest proportions since 1988 according to NASA.
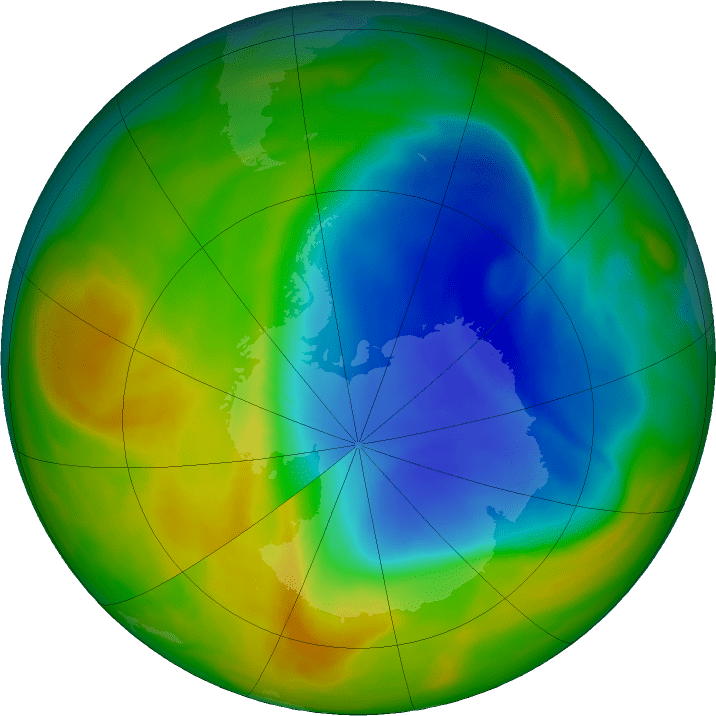
Image via NASA / Ozone Hole Watch.
Spreading up to 7,6 million square miles (19.69 million km2) at its peak this year on September 11, the hole in our atmosphere’s ozone layer seems to be healing. The hole, currently looming above Antarctica, was 1.3 million square miles (3.36 million km2) smaller than last year, the smallest it’s ever been since 1988, and it keeps shrinking.
Atmospheric sunscreen
Ozone, sometimes called trioxygen, is an allotrope (a particular structural layout) of oxygen. It’s a molecule made up of three oxygen atoms, O3, and is less chemically stable than the regular O2 molecule. Ozone is a pale-blue, toxic and quite smelly gas, but that’s ok since it breaks down into regular oxygen in the lower atmosphere. It’s also a relatively rare gas, generally formed in the upper atmosphere under the action of UV rays and the charges they generate in the lower layers of the stratosphere.
Because chemistry has a sense of humor, that same ozone layer absorbs virtually all (98% to 99%) of incoming, medium-frequency UV radiation. Awesome news if you like being alive or at least not in constant agony from radiation burns, since UV radiation can wreak havoc on organisms living on land. Exposure to higher levels of UV radiation (such as those associated with a thinned ozone layer) significantly raise the risks of developing cataract and skin cancer. Wide-scale exposure to UV levels expected in the absence of the ozone layer would drastically impact whole ecosystems at a time, and would significantly change the shape of life on Earth as we today know it.
Heavy use of ozone-damaging chemicals — in particular, clorofluorocarbons and hydrochlorofluorocarbons (CHCs and HCHCs) used in old-timey fridges and spray cans — burned a gaping hole in this layer in the 70’s and 80’s. These substances break apart in the upper atmosphere, freeing chlorine and bromine to bind with the ozone. The reactions are quicker over Antarctica (where the hole currently looms) as the frozen polar stratospheric clouds offer surfaces upon which heterogeneous chemical reactions take place.
The hole understandably had everyone quite panicked. But it’s starting to show signs of healing up.
NASA researchers believe that the increasing temperatures we’ve seen in later years helped plug the hole, as warmer bodies of air in the stratosphere help limit the rise of damaging chemicals, such as chlorine and bromine, to the ozone layer.
“The Antarctic ozone hole was exceptionally weak this year,” Paul Newman, chief scientist for Earth Sciences at NASA’s Goddard Space Flight Center in Greenbelt, Maryland, said in a statement. “This is what we would expect to see given the weather conditions in the Antarctic stratosphere.”
This effect, however, is like the jiggling of the gas indicator while your car is running — quite tiny, and quite chaotic. The lion’s share of the progress, the draining of the gas from your car’s tank in our metaphor, can be tracked down to the year 1987 when the evidence of ozone degradation grew so worrying authorities implemented the Montreal Protocol on Substances that Deplete the Ozone Layer. Over time, the protocol grew to be an international agreement which drove massive efforts, all throughout the world, to phase out most ozone-depleting chemicals.
Almost like international agreements on threatening environmental issues are a good thing in the long run and probably shouldn’t be abandoned, right?

Seen in April at the March for Science, Washington DC. Image credits Becker1999 / Wikimedia.
The good news marks the 30th anniversary of the ozone hole’s discovery. NASA further reports that the ozone hole was at its widest in the year 2000, at 11.5 million square miles (29.78 million km2) and will likely need a further 50 years to return to its 1980s size.
Overall, very encouraging news, but one that goes to show the huge inertia Earth’s systems have in relation to change. A lesson we should take to heart as we try to grapple climate change.






