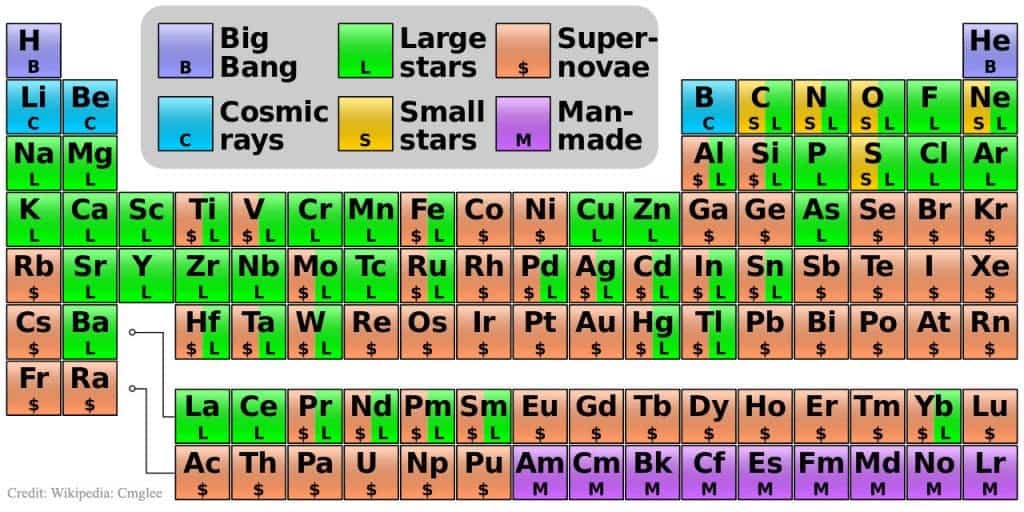
In January, four new elements were introduced to the periodic table, but they didn’t have a name. Now, they will be officially added to the periodic table, with proper names.
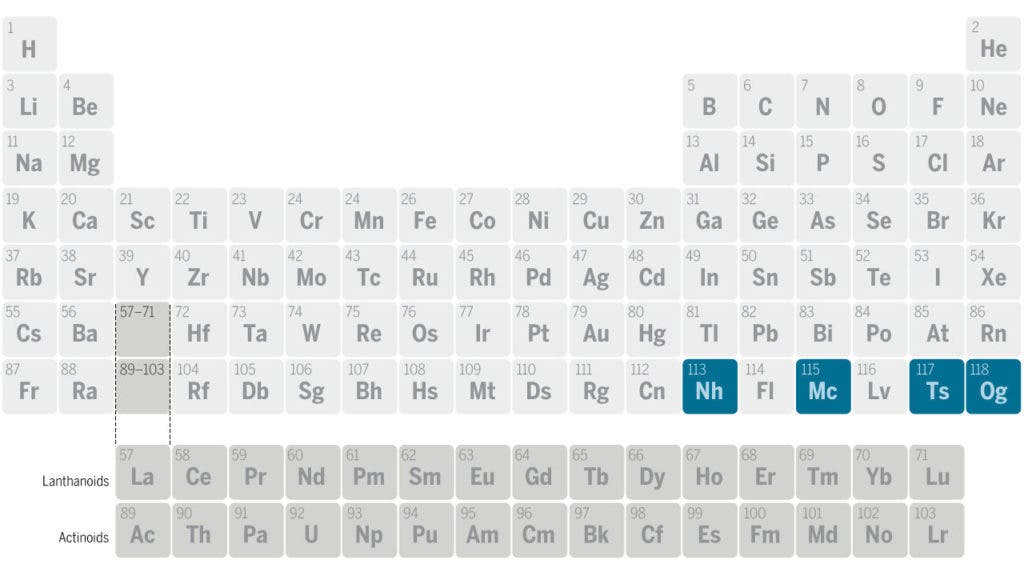
The new elements have the atomic number (Z) of 113, 115, 117 and 118 respectively. Teams of researchers from US, Russia, and Japan have decided that the elements will be called:
- Nihonium with the symbol Nh, for the element with Z =113, named after Japan.
- Moscovium with the symbol Mc, for the element with Z = 115, named after Moscow.
- Tennessine with the symbol Ts, for the element with Z = 117, named after Tennessee.
- Oganesson with the symbol Og, for the element with Z = 118, named after Yuri Oganessian, a nuclear physics professor at the Joint Institute for Nuclear Research.
“It is a pleasure to see that specific places and names (country, state, city, and scientist) related to the new elements is recognized in these four names. Although these choices may perhaps be viewed by some as slightly self-indulgent, the names are completely in accordance with IUPAC rules”, commented Jan Reedijk, who corresponded with the various laboratories and invited the discoverers to make proposals.
“In fact, I see it as thrilling to recognize that international collaborations were at the core of these discoveries and that these new names also make the discoveries somewhat tangible.”
These elements were synthesized artificially and are not found in nature. They’re only stable for extremely short periods of time, which is why we don’t know too much about them and we can’t really perform any chemical experiments with them.
But with these four additions, the last row of the periodic table is complete — but that’s not to say that we can’t add another! Researchers will likely start synthesizing new, different elements, but we’ll need to add another row for that — or rather, a new block. This is truly something exciting and groundbreaking which could open new doors in modern chemistry. But of course, we’ll have to wait for scientists to actually create new elements before we can know.