We’re one leg closer to developing functional limb regeneration.

Image credits Celia Herrera-Rincon / Tufts University.
A team of researchers from the Tufts University wishes that everyone could lose a foot and have it, too. The group successfully “kick-started” partial tissue regrowth in adult African clawed frogs (Xenopus laevis) through the use of a bioreactor and electroceutical (electrical cell-stimulating) techniques.
The cradle of life
“At best, adult frogs normally grow back only a featureless, thin, cartilaginous spike,” says senior author Michael Levin, developmental biologist at the Tufts University’s Allen Discovery Center.
“Our procedure induced a regenerative response they normally never have, which resulted in bigger, more structured appendages. The bioreactor device triggered very complex downstream outcomes that bioengineers cannot yet micromanage directly.”
The scientists split up the frog models into three groups — one experimental, one control, and one ‘sham’ group. Each animal had one of its hindlimbs amputated for the trial. Next, they 3D printed a “wearable bioreactor” out of silicon and filled it with hydrogel (a tissue-like mix of water and polymers). This hydrogel was mixed with certain silk proteins that provided a “pro-regenerative environment” and “enhanc[ed] bone remodeling”, according to the authors.
Next came the trial proper: frogs in the experimental and sham groups received the bioreactor (which was sutured on) immediately after the amputation procedure. The difference between the two is that the hydrogel for the experimental group was further laced with progesterone. Progesterone is a hormone that works to prepare the body for pregnancy but has also been shown to promote tissue repair, from nerves to bone. The control group received no treatment. Twenty-four hours later, the devices were removed.
Observations carried out at various times over the following nine-and-a-half months show that the bioreactor induced a degree of regeneration in the experimental group that had no counterpart in the other two groups. Instead of the typical spike-like structure, frogs treated with the bioreactor-progesterone combo re-grew a paddle-like structure — closer to a fully formed limb than what unaided regeneration processes created.
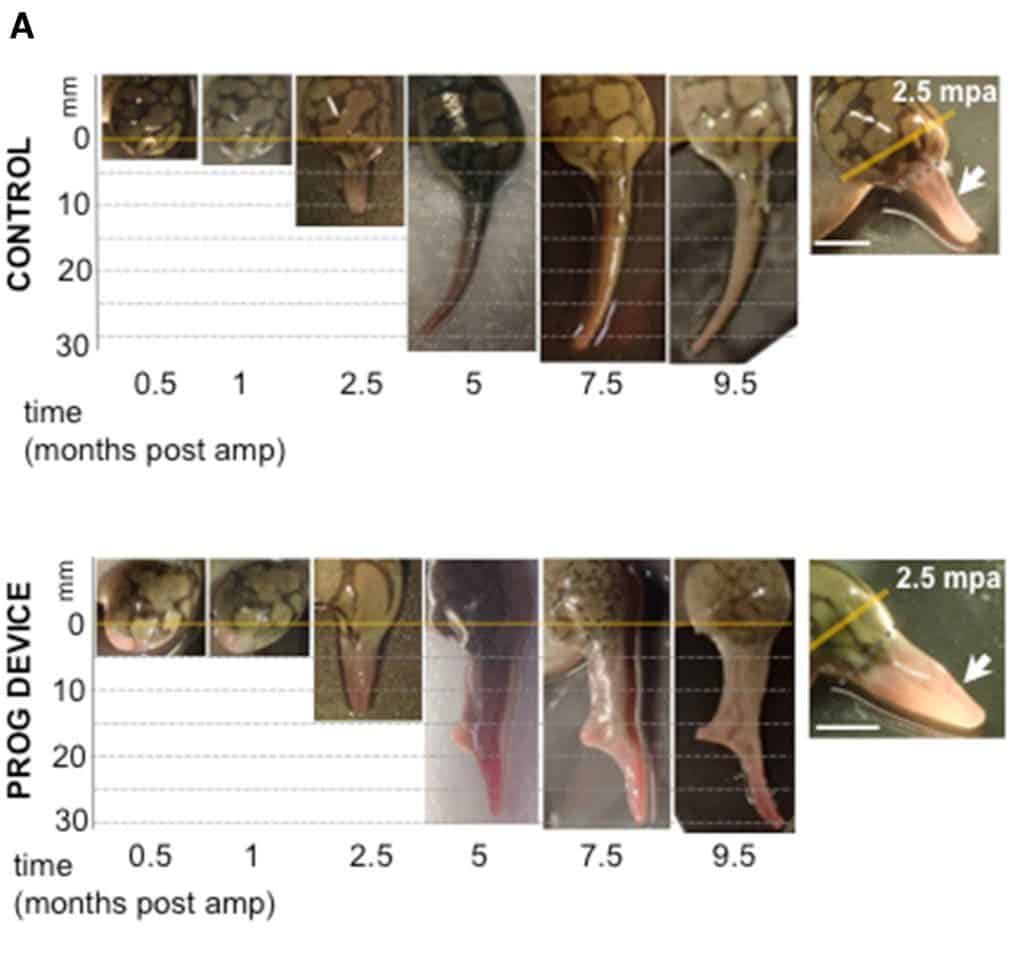
“The bioreactor device created a supportive environment for the wound where the tissue could grow as it did during embryogenesis,” says Levin. “A very brief application of bioreactor and its payload triggered months of tissue growth and patterning.”
The regenerated structures of the experimental groups were thicker, had better-developed bones, nerve bundles, and blood vessels. Video footage of the frogs in their tanks also showed that these frogs could swim more like un-amputated ones, the team adds. Scarring and immune responses were also dampened in the bioreactor-treated frogs, suggesting that the progesterone limited the body’s natural reaction to injury in a way that benefited the regeneration process.
So, why exactly did the device work? Genetic tests performed by the team showed that the bioreactor-progesterone combo altered gene expression in cells at the amputation site. Genes involved in oxidative stress, serotonergic signaling, and white blood cell activity were upregulated, while some other signaling-related genes were downregulated.
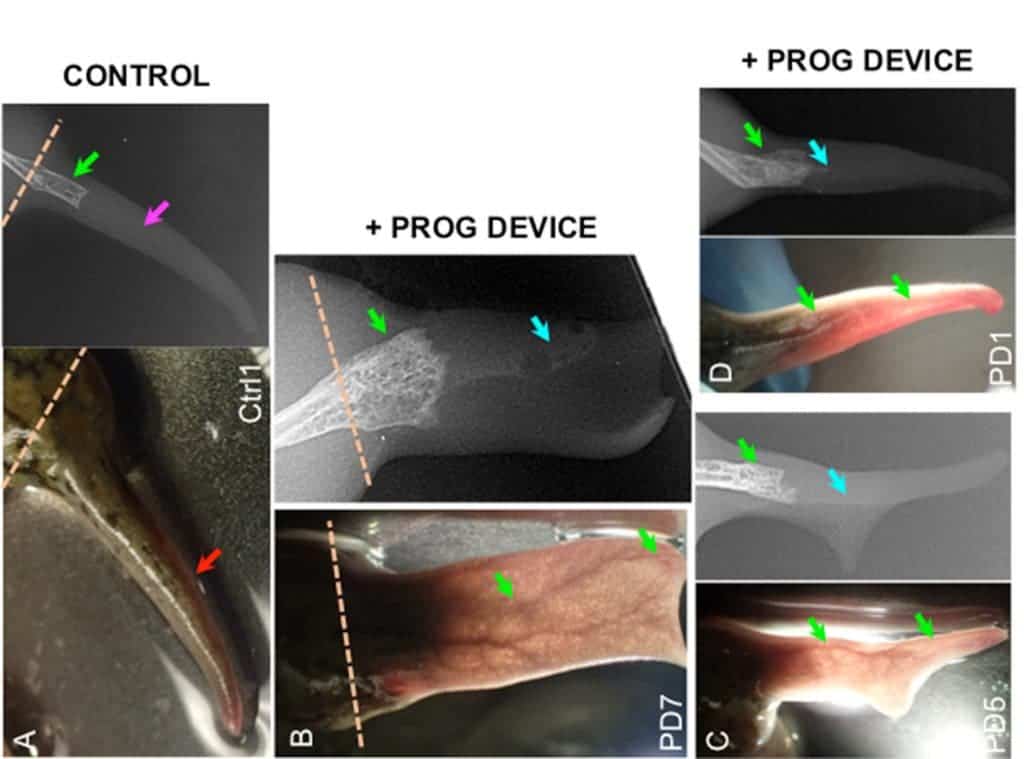
Image credits Celia Herrera-Rincon et al., 2018, Cell Reports.
“In both reproduction and its newly discovered role in brain functioning, progesterone’s actions are local or tissue-specific,” says first author Celia Herrera-Rincon, neuroscientist in Levin’s lab at Tufts University.
“What we are demonstrating with this approach is that maybe reproduction, brain processing, and regeneration are closer than we think. Maybe they share pathways and elements of a common — and so far, not completely understood — bioelectrical code.”
The team plans to expand their research in mammal subjects. Previous research hinted that mice can partially regenerate tissue (such as amputated fingertips) under the right conditions. Life on land, however, hinders this process. “Almost all good regenerators are aquatic,” Levin explains, adding that “a mouse that loses a finger or hand, and then grinds the delicate regenerative cells into the flooring material as it walks around, is unlikely to experience significant limb regeneration.” Still, let’s keep our fingers crossed that the team finds an elegant and efficient solution to this problem — it may, after all, be our limbs that we regrow one day!
But there’s much work to be done until then. Levin says the next step is to add sensors to the device for remote monitoring and optogenetic stimulation, which should give the team a degree of control over how tissues regenerate in the bioreactor. They also plan to expand on their work with bioelectric processes in the hopes of successfully inducing regeneration in the spinal cord, and to the merits of this approach for tumor reprogramming
The paper “Brief Local Application of Progesterone via a Wearable Bioreactor Induces Long-Term Regenerative Response in Adult Xenopus Hindlimb” has been published in the journal Cell Reports.






