Ahh, the phallus. In most sexually-reproductive species, half of the individuals lack one, while the other half is constantly trying to share theirs as much as possible with the first group, with varying degrees of success — bragging, fighting or impressing their way to the continuation of the species. Marvelous!

Image via wikipedia
However, for the importance it has in our lives both culturally — often associated with fertility and virility — and biologically, we don’t really know how it evolved. Did species develop it separately, as the most efficient solution to the problem of sexuality? Did we all get one from a common ancestor but some just kind-of got tired of it and developed other methods of insemination?
,
Tackling a hard problem
The evolution of internal fertilization was a critical step towards the diversity of amniotes (animals that lay eggs or nurture it inside the mother’s body) we see today. In most cases, this process relies on a critical step: fertilization requires the deposition of male genetic material in the form of sperm, facilitated by the phallus, inside the female’s cloaca (for birds) or vagina (for mammals). Other species, such as 97% of all bird species and some reptiles, rely on cloacal apposition.
One such reptile, the tuatara or Sphenodon punctatus, provided the team with the biological niche they needed to solve the case — though resembling lizards, they are acually part of a separate lineage, belonging to the order Rhynchocephalia. But there was a little problem; being an endangered species since 1895, they couldn’t use recent embryos to study the reptiles in the early developmental stages they were interested in:
“As the sister group to squamates [lizards and snakes], S. punctatus occupies an important phylogenetic position for resolving amniote genital evolution; however, acquisition of new embryological material is difficult owing to the close management of this species,” the team writes.
So they set out to look for suitable material and found four specimens that were donated to the Harvard Embryological Collection (HEC) in 1909 by Arthur Dendy, who owned the most comprehensive collection of this species. One of these specimens was just what they were looking for.
“We examined the historical records and concluded that of the four S. punctatus embryos sent to Minot [then curator of the Harvard Embryological Collection], one embryo, specimen 1491, is the ideal stage at which to examine the cloacal region for evidence of genital swellings.”
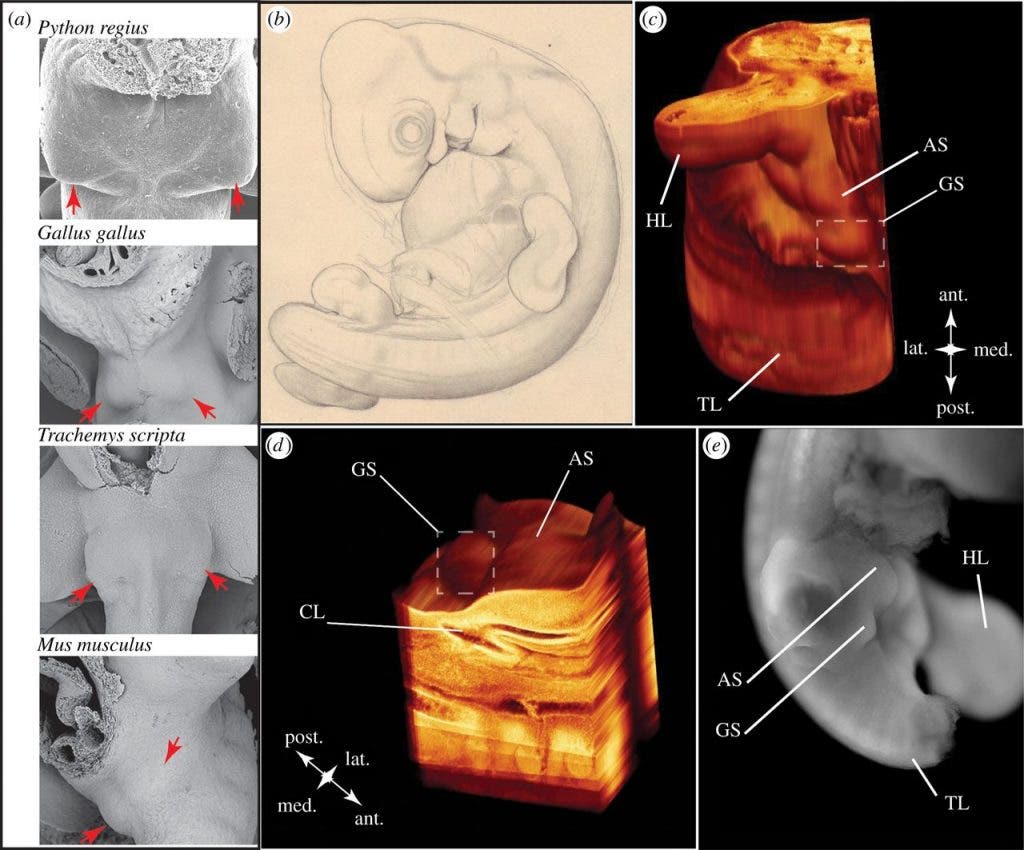
(a, red arrows) Specimens taken from indicated species.
(b) Illustration of tuatara embryo 1491 from the Harvard Embryological Collection (HEC).
(c,d) This embryo possesses genital swellings (GS) and anterior swellings (AS) adjacent to the cloaca (CL) at a stage comparable with when other amniote species are undergoing external genital morphogenesis.
Image via royalsocietypublishing, credit to authors.
Specimen 1491 was histologically prepared into 8µm thick sagittal sections, but the position of the animal’s body (image b) didn’t allow the team to study the genitals directly, so they reconstituted 82 slides into a 3D model they could analyze.
The model revealed genital swellings between the hind limbs and cloaca. Another, more focused modeling, was built to analyze only the cloacal region, to better understand the relationship between the genital swellings and the cloaca. Here, one swelling is visible right next to the animal’s urogenital opening — called the urodeum — correlating to the right genital swelling in squamates, and another one anterior to the cloaca, also correlating with squamate development in this phase.
“Thus, the reconstruction of the S. punctatus cloacal region reveals the presence of the right genital and anterior cloacal swellings, indicating that although this species lacks an intromittent phallus as an adult, development of a phallus and cloacal lip is initiated in the embryo.”
“In chicken and quail, outgrowth of paired genital swellings initiates morphogenesis of the resultant genital tubercle, which later regresses owing to programed cell death. Our data indicate that the early stages of external genital development occur in S. punctatus embryos, and we suggest that the absence of an intromittent phallus in adult tuatara could result from a similar process of programmed cell death or diminished proliferation at stages later than embryo 1491,” the team concludes.
So what does this mean? Well, embryo development follows some wacky rules. When an organism evolves a new trait, it doesn’t actually loose the genetic information is had up to then, rather it’s being deactivated — this is why atavistic characteristics are sometimes developed. As the embryo matures and grows, it goes through developmental stages that won’t necessarily be retained — such as whale and dolphin embryos developing legs, then loosing them.
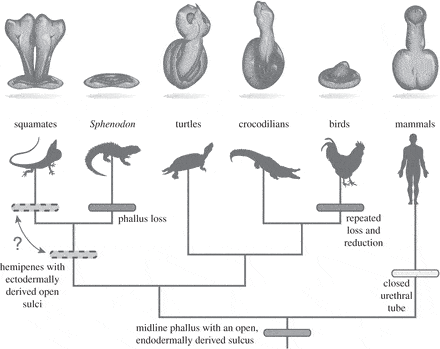
Image via royalsocietypublishing, credit to authors.
As such, the presence of the genital swellings in an animal that lacks a phallus as an adult suggests that the organ was inherited from one common ancestor, and then developed into all the variations we see today.
“Taken together with previous studies of external genital development, the data from tuatara support the hypothesis that the amniote phallus had a single evolutionary origin that was followed by lineage-specific modifications that underlie the diversity observed in extant amniotes.”






