It’s generally accepted among physicists that the solar system formed through the gradual accumulation of rocky material. Over a hundred million years or so, planets would form out of this primordial, chaotic dance of colliding rocks. Zooming in on this process, however, shows that there are still many mechanisms still unaccounted for. But we are learning more. Two newly published paper help shed light on a confusing matter, showing how it’s possible for the planetesimals that went on to form Earth to melt and evaporate material.
Up in smoke
Ancient meteorites called chondrites are thought to reflect the chemistry of the early solar system. Astronomers study them with a keen eye because they know they might hold key insight into how planets like Earth or Mars formed billions of years ago. But there’s also a caveat. If Earth and other rocky planets in the solar system should be similar to chondrites, why are the latter so rich in more volatile rock-forming elements?
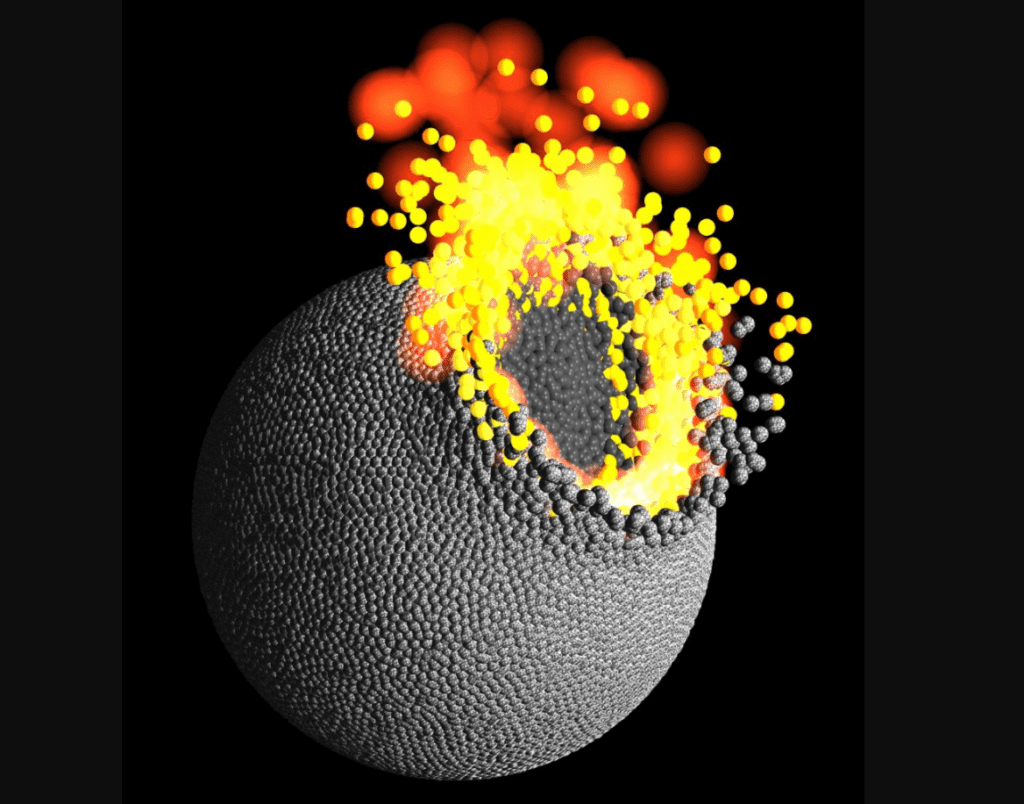
New research suggests Earth’s chemical composition may have been shaped by the evaporation of molten rock following violent collisions with planetesimals. These are objects made from dust, rock, and other materials, which range in size from several meters to hundreds of kilometers.
According to one theory put forth by Russian astronomer Viktor Safronov, a planetary system forms from a disk of material seeded by a nebula. Under the influence of gravity, material will accrete into small chunks that get larger and larger forming planetesimals. Many such objects will break apart as they collide into each other but others will grow eventually becoming planets and moons. That’s why it’s so important to mention planetesimals when discussing solar system formation.
Earth may have lost nearly 40 percent of its mass as vapor during collisional growth
In one recent paper published in Nature, a team led by Remco Hin and colleagues at the School of Earth Sciences, University of Bristol, UK, looked at the isotope ratios of magnesium for Earth, Mars and some asteroids. Their analysis found Earth and similar rocky planets are enriched in the heavier isotope 25Mg compared to chondrites from which they’re supposedly formed. This is what you’d expect to see as a consequence of the evaporation of molten rock from growing planetesimals.
Hin says these results mean that Earth lost nearly 40 percent of its mass as vapor during collisional growth.
“When I first saw the result that about 40 percent of a planet’s mass is lost as vapour, I could hardly believe them and thought I must have made a calculation error somewhere. After checking, double-checking and triple-checking, the result stayed the same. I briefly went into denial refusing to believe that a planet could lose nearly half its mass during its growth, until I finally asked myself the question what argument I had against believing it. I had none. So, faced with various rational arguments in favour of thinking it was possible, and none against it, I caved in and thought it had to be right,” Hin wrote to me in an e-mail.
During one such violent collision, the Earth (in its current form) and the moon were formed — but this happened a later stage of the planet’s evolution, when the Earth had enough gravity to keep the vapor from escaping into outer space. ” Smaller bodies have lower gravity and so vapor is lost to space. Thus, the bodies as a whole acquire heavy isotope ratios (Mg in the case we studied),” Hin said.
In an accompanying paper by Oxford University scientists, a team led by Bernard Wood recreated similar conditions seen during Earth’s formation by melting rock in a furnace at 1,300 degrees Celsius in oxygen-poor conditions. Their results show some elements could escape in vapor released from the molten rock in proportions similar to that observed within Earth.
“Our experiments indicate that the pattern of volatile element depletion in the Earth was established by reaction between molten rock and an oxygen-poor atmosphere. These reactions may have occurred on the early-formed planetesimals which were accreted to Earth or possibly during the giant impact which formed the moon and which is believed to have caused large-scale melting of our planet,” Wood said in a press release.
Together, the two papers exemplify a proven process that can explain why Earth is fairly depleted of certain elements that are abundant elsewhere in the solar system. Considering there are many competing planetary formation models, this new insight could be key to achieving a more accurate picture of how the early solar system looked like.
It’s a bit too early to generalize, though. Hin tells us that we still need more data to model the chemical compositional changes following vapor loss.
“The input data we used in our chemical model are, in my opinion, the best available to date, but they can be still be improved a lot over the next years. To know whether the process of vapour loss from small planetary bodies during their growth occurs universally we also need to further explore the underlying physics. We have made a start with this in our publication, but firmer conclusions require a more detailed model,” Hin said.
Next, researchers from Wood’s group plan on studying the volatility of other elements for this process, like chlorine and iodine. Hin and colleagues, on the other hand, are now in the process of securing more data to verify their model.






