Japanese researchers at the University of Kyoto have recently demonstrated a porous polymer that selectively binds to carbon dioxide molecules. It is ten times more efficient than similar other materials and is made from inexpensive materials. In the future, such a material could be incorporated into the exhausts of fossil fuel power generators or carbon capture stations. The CO2 would not only be prevented to reach the atmosphere, where it raises temperatures, it could also be turned into a useful product.
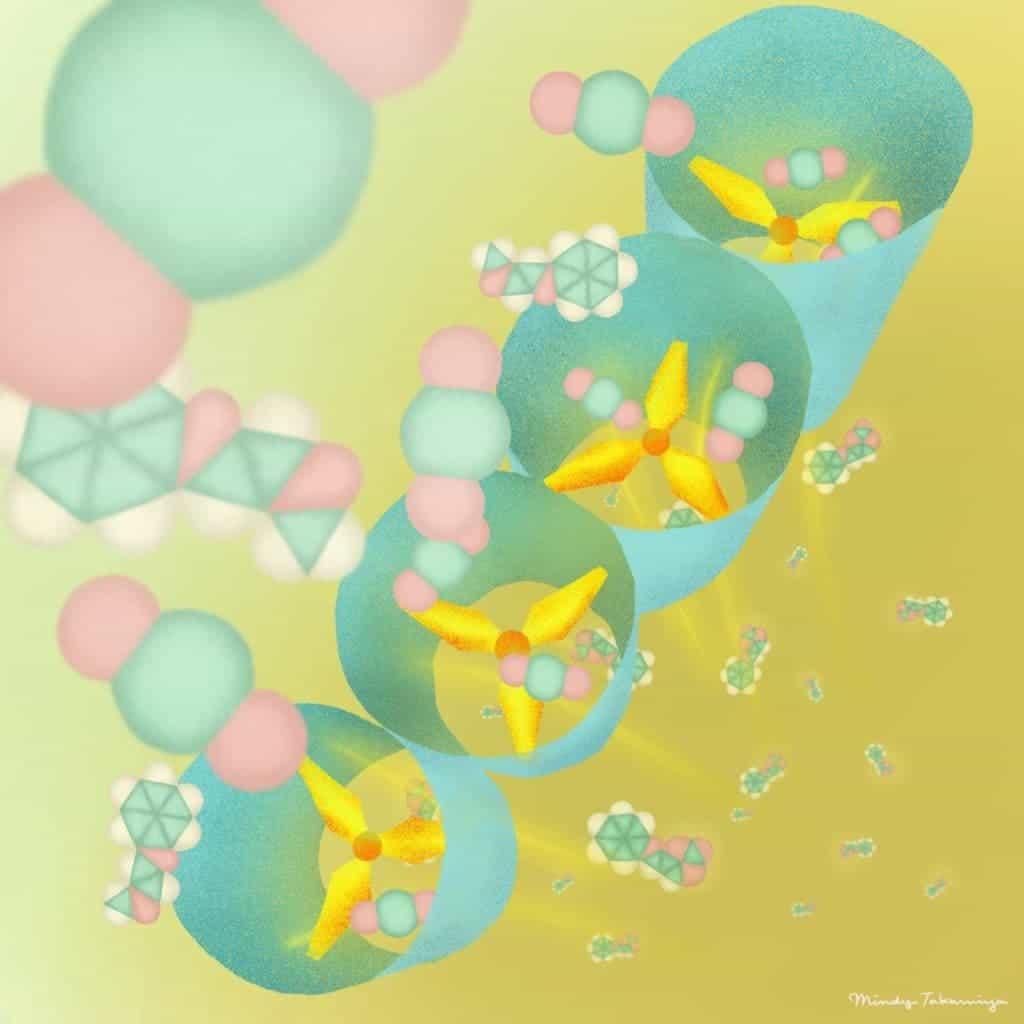
The new material belongs to a class called porous coordination polymer (PCP), also known as metal-organic frameworks (MOF). It mainly consists of zinc metal ions and an organic component, known as a ligand, with a propeller-like molecular structure.
When CO2 molecules approach the structure, the ligands rotate and rearrange themselves, thereby trapping the carbon. This results in slight changes in the molecular channels of the PCP, which basically acts as a sieve.
X-ray structural analysis revealed that the material only interacts with carbon dioxide molecules, which are captured 10 times more efficiently than other PCPs.
No energy input is required for the process to occur because it is favorable for the CO2 to bind to the zinc ions. Once bound, the CO2 molecules become activated and capable of reacting with other molecules.
“We have successfully designed a porous material which has a high affinity towards CO2 molecules and can quickly and effectively convert it into useful organic materials,” says Ken-ichi Otake, a materials chemist at Kyoto University.
The researchers claim that the CO2 can be incorporated into useful organic materials, such as cyclic carbonates which can be used in petrochemicals and pharmaceuticals.
The findings appeared in the journal Nature Communications.



