The tree of life consists of three domains: Archaea, Bacteria, and Eukarya. The first two are all prokaryotic microorganisms, or single-celled organisms whose cells have no nucleus. Eukarya can be both unicellular and multicellular organisms. But what did the very first life forms look like? Scientists suspect that Bacteria and Archaea evolved from a hypothetical Last Universal Common Ancestor, or LUCA for short, because the organism’s cell membrane was an unstable mixture of lipids.
Dutch researchers at the University of Groningen engineered an E. coli bacterium whose membrane is both a mix of Bacteria and Archaea. They found that this mixed membrane was stable, refuting the main hypothesis for the ‘lipid divide.”
A mixed membrane new life form

A bacteria’s membrane is composed of straight-chain fatty acids that are ester-linked to a backbone of glycerol-3-phosphate. In Archaea, the backbone is glycerol-1-phosphate, to which isoprenoids are linked by ether bonds. According to the ‘lipid divide’ hypothesis, a mixed membrane of phospholipids would be less stable than a homogeneous membrane of just one type of phospholipid. “So eventually a split occurred, resulting in the two domains of Bacteria and Archaea,” commented Arnold Driessen, who is a Professor of Molecular Microbiology at the University of Groningen.
This split — if it ever happened — must have occurred 3.5 billion years ago. Alas, this primeval event has not been recorded in the fossil record, which left Driessen and colleagues with only one viable option: reverse-engineer a microorganism with a mixed membrane.
The University of Groningen researchers were not the first to attempt this endeavor. However, the previous trials engineered bacteria with no more than 1% archaeal lipids. Using a novel approach, Driessen and colleagues made a huge leap by devising a viable cell comprised of a staggering 30% archaeal lipids.
“The main challenge was to get E. coli cells to synthesize significant amounts of ether lipids. In the work there were two key elements, introduction in addition to the enzymes of the ether lipid pathway, the by us recently identified archaeol synthase; and the genomic duplication of the E. coli MEP/DOXP+ pathway to stimulate isoprenoid biosynthesis which is normally is at a low level. When both were brought together this led to the biosynthesis of significant amounts of ether lipids in E. coli and this was the ‘Aha’ moment!” Driessen told ZME Science.
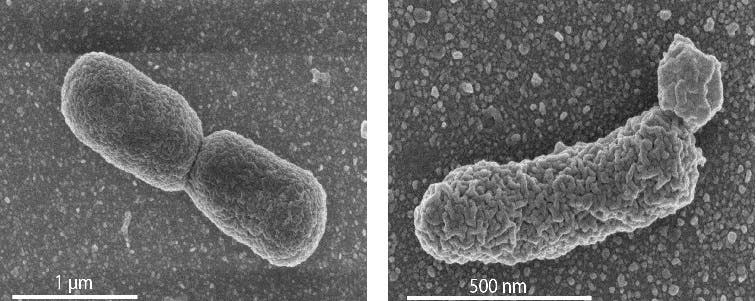
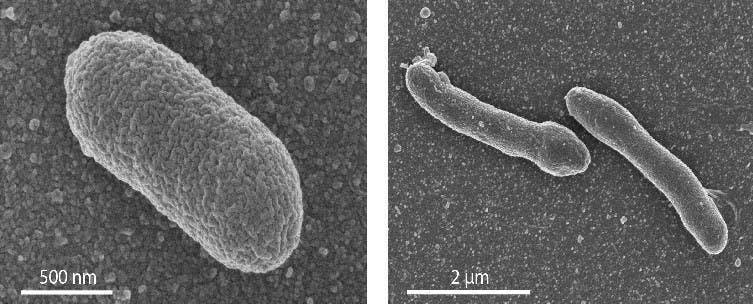
In the newly engineered E. coli, the phosphatidylglycerols — the lipids which form the basic bilayer of the bacterial membrane — were replaced by their archaeal equivalent (archaetidylglycerol). Despite this transformation, the bacterium grew normally and was stable, which refutes the hypothesis that a mixed membrane is inherently unstable. The new life form also exhibited some differences from the unmodified E. coli, such as a more elongated cell.
Although it refutes the ‘lipid divide’ hypothesis, the research does offer some clues as to what may have driven a Bacteria/Archaea divide. For one, the archaeal enzymes that are crucial for archaeal membrane lipids are less specific in the reactions they catalyze than the bacterial counterparts. Driessen believes enzyme specificity could have one of the drivers of the divide.
In any case, it’s a challenging inquiry because the reverse-engineering process is far from perfect. For one, the researchers modified a modern E. coli bacteria — the product of countless generations that evolved over the course of 3.5 billion years. When I asked Driessen how far he thinks scientists are from modeling a primordial organism, he wasn’t very optimistic — “surely far away from the primordial form, as the primordial organism had a completely different protein make up,” he said.
“The thing we mimic, if you can say so, is a mixed heterochiral membrane, and our data shows that this can lead to a stable organism suggestion that the lipid divide was not driven by an intrinsic instability of such membranes. Of course, this was a test with a system that evolved into what it is now, thus with membrane proteins that are used to function in a homochiral membrane. Apparently, these membrane proteins function readily is such ‘primordial’ membranes,” Driessen continued.
Next, the researchers plan on engineering an E. coli bacterium which relies entirely on archaeal ether lipids.
“We would like to replace the entire phosphatidylethanolamine (PE) pool for archatidylethanolamine (AE), to make an E. coli which relies entirely on archaeal ether lipids. However, this is far from trivial. PE is a non-bilayer lipid, AE shows a much more complex phase behavior, so it may not compensate for the polymorphic behavior of PE which is a requirement for viable E. coli,” Driessen said.
“Synthetic biology may be able to construct presumed primordial life forms such that evolution can be recreated in the laboratory so that we can start to test/challenge evolution theories. It is a challenge for an experimental evolutionary molecular biologist, but need to step away from the theory and validate hypotheses experimentally.”
Scientific reference: Antonella Caforio, Melvin Siliakus, Marten Exterkate, Samta Jain, Varsha Jumde, Ruben Andringa, Servé Kengen, Adriaan Minnaard, Arnold Driessen, John van der Oost, Converting Escherichia coli into an ‘archaebacterium’ with a hybrid heterochiral membrane, Proceedings of the National Academy of Sciences, 2018.






