The new material is eerily life-like.
What makes something alive? Is it the fact that it has a metabolism, that it can organize itself into a coherent structure? If that’s all it takes, then researchers might have just created artificial life. A Cornell team took advantage of some of DNA’s unique properties to develop a life-like material that can self-organize, self-assemble, and even metabolize nutrients.
“We are introducing a brand-new, lifelike material concept powered by its very own artificial metabolism. We are not making something that’s alive, but we are creating materials that are much more lifelike than have ever been seen before,” said Dan Luo, professor of biological and environmental engineering in the College of Agriculture and Life Sciences.
DNA is the foundation of all life on Earth. It contains the instructions needed for an organism to survive and develop, producing new cells and sweeping old ones away in a hierarchical pattern. However, DNA is also a polymer, meaning it has some useful bio-construction properties that researchers can use.
In this study, Luo and colleagues used what they call DASH (DNA-based Assembly and Synthesis of Hierarchical) materials to create a biomaterial that can autonomously emerge from its nanoscale building blocks and arrange itself — first into simple polymers, then into more complex shapes.
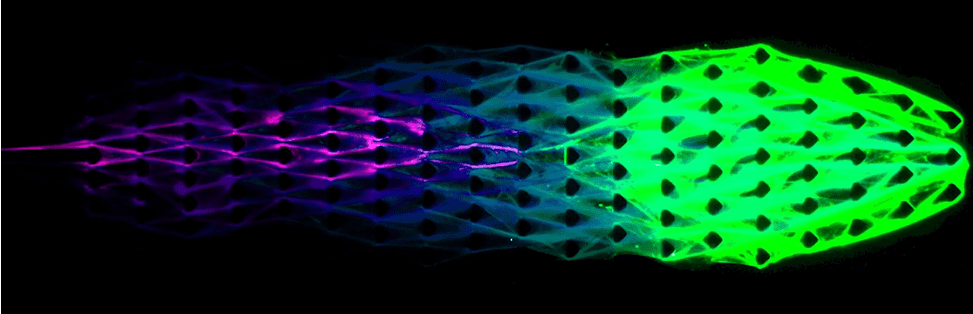
They started from a sequence of 55 nucleotides (the building blocks of DNA and RNA) and from there, the DNA molecules were multiplied hundreds of thousands of times, creating chains of repeating DNA reaching a few millimetres in size. Then, this reaction was injected into a microfluidic device that the necessary energy and materials for biosynthesis.
As material gathered more and more resources, the DNA was able to synthesize new strands at the front end, while the tail end degraded to maintain an optimum balance. Using this mechanism, it was also able to move around, even against the flow — very similar to how slime molds move.
“The designs are still primitive, but they showed a new route to create dynamic machines from biomolecules. We are at a first step of building lifelike robots by artificial metabolism,” said Shogo Hamada, lecturer and research associate in the Luo lab, and lead and co-corresponding author of the paper. “Even from a simple design, we were able to create sophisticated behaviors like racing. Artificial metabolism could open a new frontier in robotics.”
If that wasn’t life-like enough, researchers are currently working on ways to improve longevity and self-replication.
“Dynamic biomaterials powered by artificial metabolism could provide a previously unexplored route to realize “artificial” biological systems with regenerating and self-sustaining characteristics,” the study concludes.
The goal is not to produce artificial life, but rather to use the system as a biosensor or as a dynamic template for making proteins without living cells.
The study was published in Science Robotics.






