A rather unexpected finding might improve our understanding of how clouds form and develop.
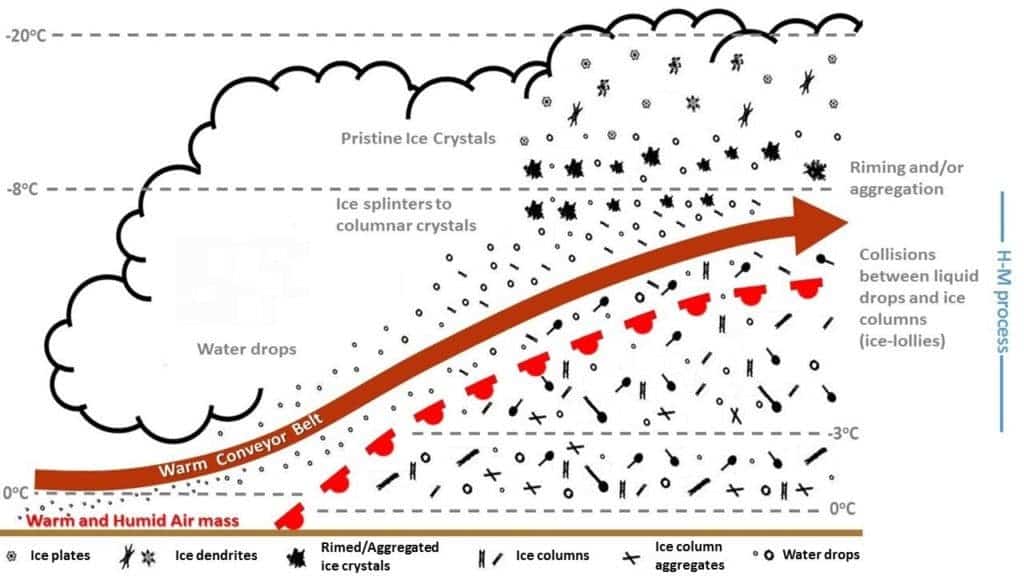
Clouds filled with lollipop crystals sounds like a drug-induced vision, but it’s actually what researchers found after analyzing more than 5 million images taken during a 2009 flight across England.
“Natural processes can create objects of real beauty,” said Jonathan Crosier, a senior research fellow who studies clouds at the University of Manchester, and one of the scientists involved in the work. “We instantly started asking ourselves questions about how they form.”
Crosier and his colleagues were looking for things that would help them learn more about the precipitations and the lifetime of clouds. They were quite surprised to see numerous crystals which looked like lollipops — a spherical part on top of a “stick.” It wasn’t the first time these crystals were observed, but it was the first time they were observed in such a large number, enough to indicate that they weren’t some kind of accident.
In order to understand them, we first have to understand that some clouds exist in a mixed-phase — they contain both solid ice and liquid super-cooled water (below freezing temperature, but still liquid). These clouds exist in a very delicate balance, Crosier explains.
“Too much ice and the ice can consume the liquid water, which can lead to precipitation and the dissipation of the cloud,” he said. “Too little, and the ice will fall out, leaving behind a highly reflective super-cooled liquid cloud, which can persist for a long time, whilst generating virtually no precipitation.”
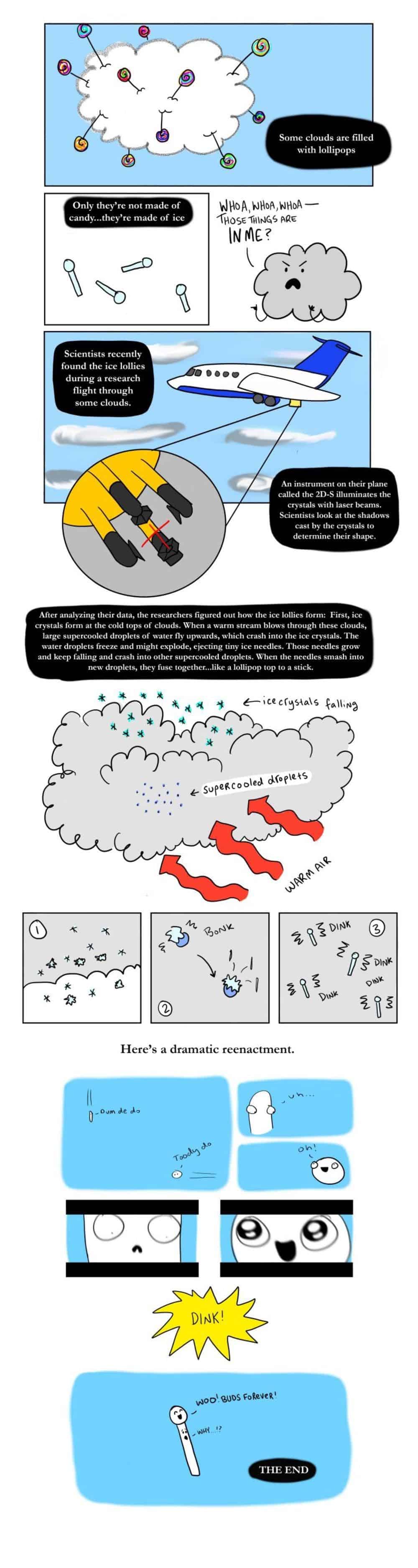
The formation of the ice lollipops happens in this type of cloud. Whenever supercooled water touches ice, it turns into ice itself. So what happens sometimes is that a warm updraft picks up these droplets of supercooled water, elongates them, and crashes them with ice crystals, causing them to freeze. So you end up with ice needles. Occasionally, these needles might break off, fall down, connect with other, unmoved and round droplets, causing these to freeze as well — creating the lollipop shape.
The ice-lollies tend to be between 0.25 mm and 1.5mm in length. They fall to the ground at around 3.6 kilometers per hour and require very specific conditions to form. Also,
“Since they were found at an altitude of around 4 kilometers, it would take just over an hour for them to reach the surface if they remain unchanged,” Crosier said. “However, when the air temperature increases above zero Celsius, then the lolly will melt and form a small rain drop. Even if the temperature profile of the atmosphere is negative — below zero Celcius — at all layers, ice-lollies might not reach the ground in their original shape, or be deformed due to other processes occurring in the clouds,” he said. And, if temperatures stay below zero Celsius, but humidity drops below 100 percent, “then the particles will start to sublimate and could vanish without a trace.”
If all this seems a bit weird, researchers also created this very cute graphic that explains things.
Journal Reference: S. Ch. Keppas, J. Crosier, T. W. Choularton, K. N. Bower — Ice lollies: An ice particle generated in supercooled conveyor belts. DOI: 10.1002/2017GL073441