Sleep might play a much more central part in memory formation, new research reveals. Sleeping allows two different brain regions involved in the process to communicate and sync.

This paper draws on previous research by the study’s senior author György Buzsáki, M.D., Ph.D. and Biggs Professor of Neuroscience at the New York University. It focuses on the hippocampus, a brain structure suspected to take part in forming permanent memories during sleep. Dr. Buzsáki found that neurons in the hippocampus fire in high-frequency bursts of activity (which he christened “ripples”) during sleep, suggesting the cells were indeed involved in memory formation.
Now, a team led by Buzsáki wants to delve deeper into the backstage of human memory.
Sleeper service
The team invented a novel brain imaging technology, the NeuroGrid, to use in their study. It consists of a collection of tiny electrodes linked together to form a sheet, that is then laid across an area of the brain. Each electrode will then continuously monitor the activity of a set of neurons, allowing the team to take a wider but highly detailed snapshot of activity in the brain.
“This particular device allows us to look at multiple areas of the brain at the same time,” said Jennifer Gelinas, M.D., Ph.D., assistant professor at Columbia University and co-first author of the paper.
Using this electrode grid (which was supplemented with additional tracking neurons implanted in deeper areas of the brain,) the team examined neural activity in several parts of rats’ brains during NREM (non-rapid eye movement) sleep, the longest stage of sleep.
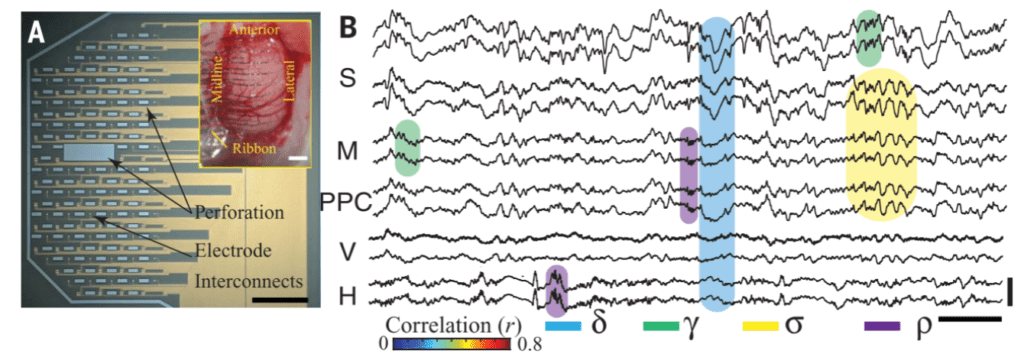
Image credits Dion Khodagholy et al., 2017, Science.
The team first confirmed the existence of the ripples Buzsáki identified in the hippocampus during sleep and also found them in certain areas of the association neocortex, a brain region involved in processing complex sensory information. A surprising find was that the ripples seemed mirrored throughout the brain, occurring in the association neocortex and hippocampus at the same time. This suggests that the two regions communicated as the animals slept.
The association neocortex has been tied to memory storage, so the team believes that this dialogue between the two structures helps the brain retain information.
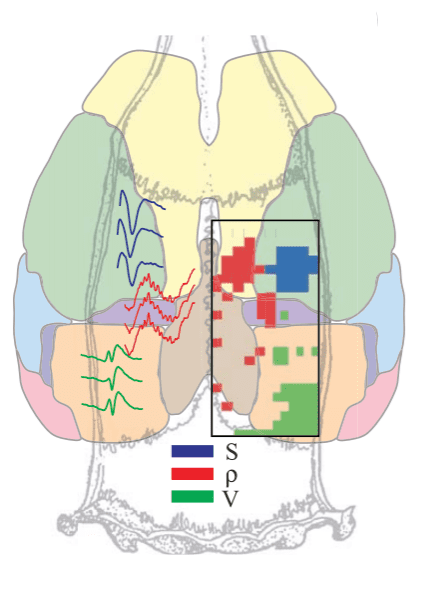
Image credits Dion Khodagholy et al., 2017, Science.
Finally, to put their theory to the test, the team trained one group of rats to locate rewards in a maze. A control group was set up, whose rats would also try to explore the maze for treats, but without any prior training — i.e. they would explore in a random fashion, without any prior memory of the maze’s layout or treat location. Afterwards, the scientists used their initial method to examine the brain activity of both groups during NREM sleep.
In the control group, these ripples in the hippocampus and cortex remained relatively constant before and after the exploration task. For the trained group, however, they report increased cross-talk between the two brain areas as well as greater synchronization (coupling) of the ripples. A second training session increased communication even more, lending weight to the hypothesis that it is fundamental to the creation and storage of memories.
“Hippocampal-PPC [posterior parietal cortex] ripple coupling increased during postlearning sleep compared to postexploration sleep, a trend that was consistent across all six trained rats,” the authors note.
“Furthermore, multiple consecutive sessions of exploration in the control rats did not induce a change in hippocampal-PPC ripple coupling, and these coupling values were significantly less than those in trained rats.”
In the future, the team hopes brain surgery patients will allow them to use the NeuroGrid to check if the same ripples form in the human brain. Another avenue of research will be to see if altering these signals in animal brains can suppress (or maybe boost) memory formation and storage — which would confirm that the ripples underpin these processes.
“Identifying the specific neural patterns that go along with memory formation provides a way to better understand memory and potentially even address disorders of memory,” Dr. Gelinas concluded.
The paper, titled “Learning-enhanced coupling between ripple oscillations in association cortices and hippocampus” has been published in the journal Science.






