Case Western Reserve University and University of Kansas Medical Center researchers recently report they’ve devised a neural prosthetic that partially restored brain connectivity in rats whose brains were injured. Effectively, the rats regained their functional behavior and were able to perform tasks similarly to normal rats, something that otherwise wouldn’t had been possible. The research gives hope that the interface may be implanted in patients suffering from severe brain trauma, consequent of accidents, war, neuro-degenerative diseases or strokes, and help them live a normal life.
Some 1.5 million people in the U.S. alone suffer from traumatic brain injuries (TBI), while there are a reported 800,000 stroke victims. These patients have neural functions impaired and have great difficulty leading a normal life, either suffering from motor weakness, psychiatric disorders or even paralysis. Most of these issues are due to poor or even lack of connectivity between some parts of the brain. Remember the alien hand syndrome?
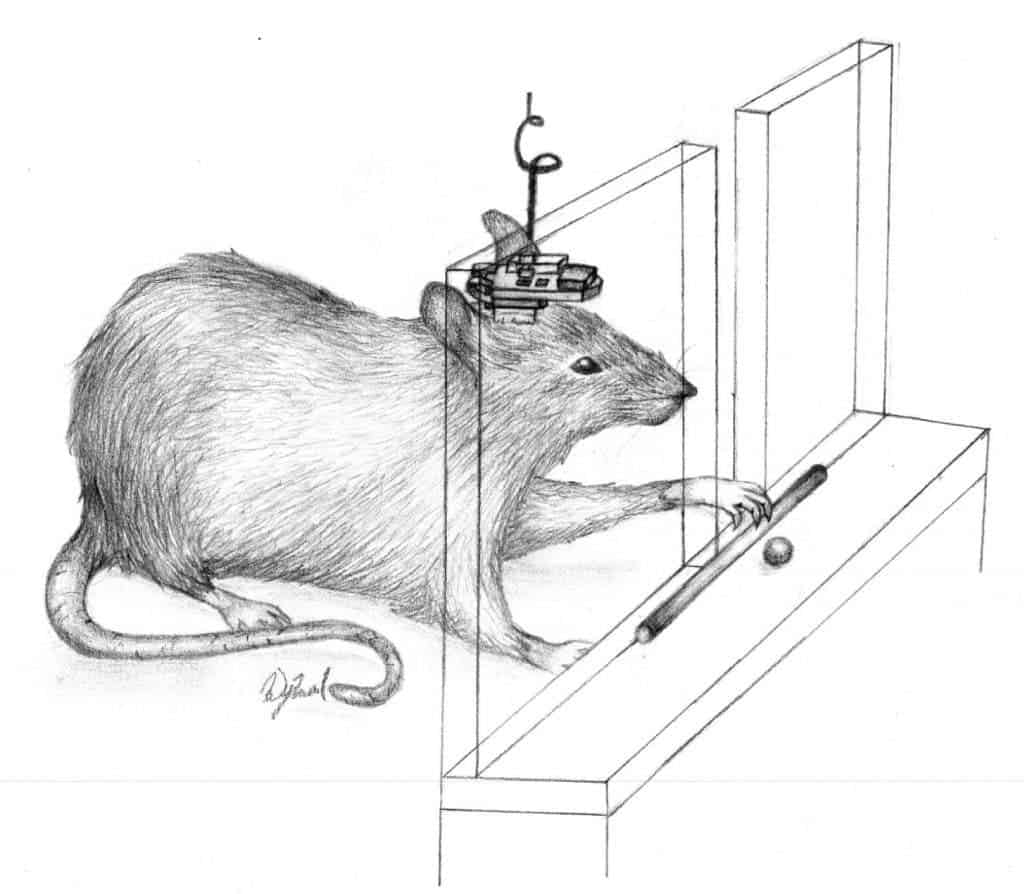
The team of researchers, led by Randolph J. Nudo, professor of molecular and integrative physiology at the University of Kansas, devised a closed-loop microelectronic system called a brain-machine-brain interface (BMBI) that bridges the areas of the brain that had lost connectivity, stimulating recovery.

Their device was tested on rat models, whose brains were mapped out. The researchers disconnected the anterior and posterior parts of the brain that control the rat’s forelimbs and then implanted at the microelectronic system atop of the animals’ heads. The electrodes of the device, which is smaller than a quarter, were carefully implanted in the two parts of the brain.
Making rat brains whole again
The BMBI works by first recording and amplifying neural signals from the anterior (front) of the brain. These signals come in various forms, and an embedded algorithm processes signal spikes and separates them from useless signals like noise. The device then sends an electrical current corresponding to each neural spike to the posterior part of the brain, stimulating neurons and effectively re-connecting the two parts of the brain.
After using the prosthetic for two weeks continuously, the researchers found that the rat models had recovered nearly all function lost due to injury, successfully retrieving a food pellet close to 70 percent of the time, or as well as normal uninjured rats. Control subjects that received random stimuli retrieved that pellets 50 percent of the time, while disabled rats who received no neural prosthetic whatsoever only managed 25 percent of the time.
The results look truly promising, and if these can be recreated in human subjects as well, then a slew of mind-numbing disorders could be addressed.
“This technology could have direct clinical application for restoring neural communication in the brain, and thus, for restoring function,” Nudo explained to KurzweilAI in an email interview. “Initial clinical targets will include focal stroke or traumatic brain injury (TBI) for motor impairments.
“However, this technology may be effective for a variety of neurological syndromes that are thought to be related to disruption of cortical communication, such as disconnection syndromes. This could include applications for cognitive and psychiatric disorders, as well. From a practical standpoint, it is theoretically possible that the target locations for the technology could be identified using modern neuroimaging techniques, such as diffusion tensor imaging, or resting state connectivity.
What’s interesting is that the there’s a sound possibility that such a system doesn’t necessarily have to be kept connected to the brain for life. It could be used for a certain time frame, after which the brain would be able to resolve its connectivity by itself. More work needs to be done, according to the authors, in order to see how long these time windows need to be and how effective recovery is post-device extraction.
“The innovation can be commercialized immediately, from the standpoint of initiating further development of a human device, navigating the FDA process for submitting an investigational device exemption (IDE), and identifying the most appropriate clinical population for first-in-human studies,” Nudo said. “Realistically, the pace of this process is constrained only by available financial resources. But, first-in-human studies could potentially take place in the 5–10 year time frame.”
A brain prosthetic

Of course, this isn’t the first brain-computer interface. Previously, ZME Science reported on a slew of mindblowing innovations. The authors of the paper published in the journal Proceedings of the National Academy of Sciences explain how their closed-loop system is different.
“This brain-machine-brain interface (BMBI) neuroprosthesis is among the first examples of closed-loop neural interfaces that combine neural recording, neural signal processing and neuromodulation functions for real-time, bidirectional interfacing with the central nervous system, and is the first such example that is applied to facilitate rapid and substantial recovery from TBI,” said Pedram Mohseni, professor of electrical engineering and computer science at Case Western Reserve.
“Other technologies that have been used to modulate brain activity in neurological conditions have been open-loop approaches (epidural stimulation, transcranial magnetic stimulation, transcranial direct-current stimulation). In addition, open-loop modulation is typically paired with rehabilitative therapy.
“In clinical trials, the rehabilitative therapy is often found to be the main factor, and not the neuromodulation technology. With this new closed-loop microdevice, at least in the rat model of TBI, no rehabilitation was required. The ‘training’ was induced in the neural pathway that was the target of the microdevice.”






