Researchers from the Massachusetts Institute of Technology (MIT) have found a way to enlarge and map brain samples. This inexpensive technique will now allow scientists to get a much closer look at the human brian and perhaps figure out some of its long standing secrets.
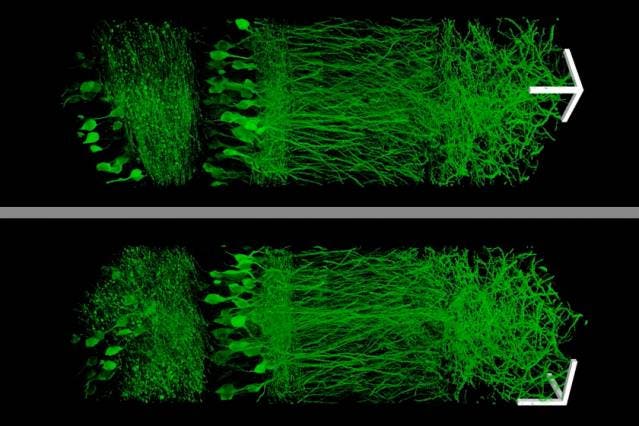
Image: Fei Chen and Paul Tillberg
Ever since the 1500s when microscopes were first developed, people have wanted to zoom in more and more. But scientists working at MIT decided to take a different approach, and instead of zooming in with the microscope, they simply made the samples bigger. Of course, that’s nowhere near as easy as it sounds. They did this by embedding samples in a polymer that swells when water is added. This allows specimens to be physically magnified, and therefore imaged at a much higher resolution.
What makes this technique even more interesting is the fact that it uses relatively cheap materials. The chemicals and materials required are commonly available everywhere.
“Instead of acquiring a new microscope to take images with nanoscale resolution, you can take the images on a regular microscope. You physically make the sample bigger, rather than trying to magnify the rays of light that are emitted by the sample,” says Ed Boyden, an associate professor of biological engineering and brain and cognitive sciences at MIT.
The thing is, with conventional microscopes, there is a physical limit to how much you can magnify. Microscopes use lenses to focus light emitted from a sample into a magnified image. However, when you get to things which are comparable in size to the wavelength of light, things start to get tricky – and you actually can’t magnify things smaller than that wavelength. When scientists do want to image at this level, they use electron microscopes. This is a big problem.
“Unfortunately, in biology that’s right where things get interesting,” says Boyden, who is a member of MIT’s Media Lab and McGovern Institute for Brain Research. Protein complexes, molecules that transport payloads in and out of cells, and other cellular activities are all organized at the nanoscale.
Scientists have tried several approaches to get past this limit, but this is the first time the sample was focused, instead of the microscopy technique. The idea is to embed samples in an expandable polymer gel made of polyacrylate, a very absorbent material found in many common products, such as diapers.
“Once the tissue is labeled, the researchers add the precursor to the polyacrylate gel and heat it to form the gel. They then digest the proteins that hold the specimen together, allowing it to expand uniformly. The specimen is then washed in salt-free water to induce a 100-fold expansion in volume. Even though the proteins have been broken apart, the original location of each fluorescent label stays the same relative to the overall structure of the tissue because it is anchored to the polyacrylate gel”, MIT’s press room writes.
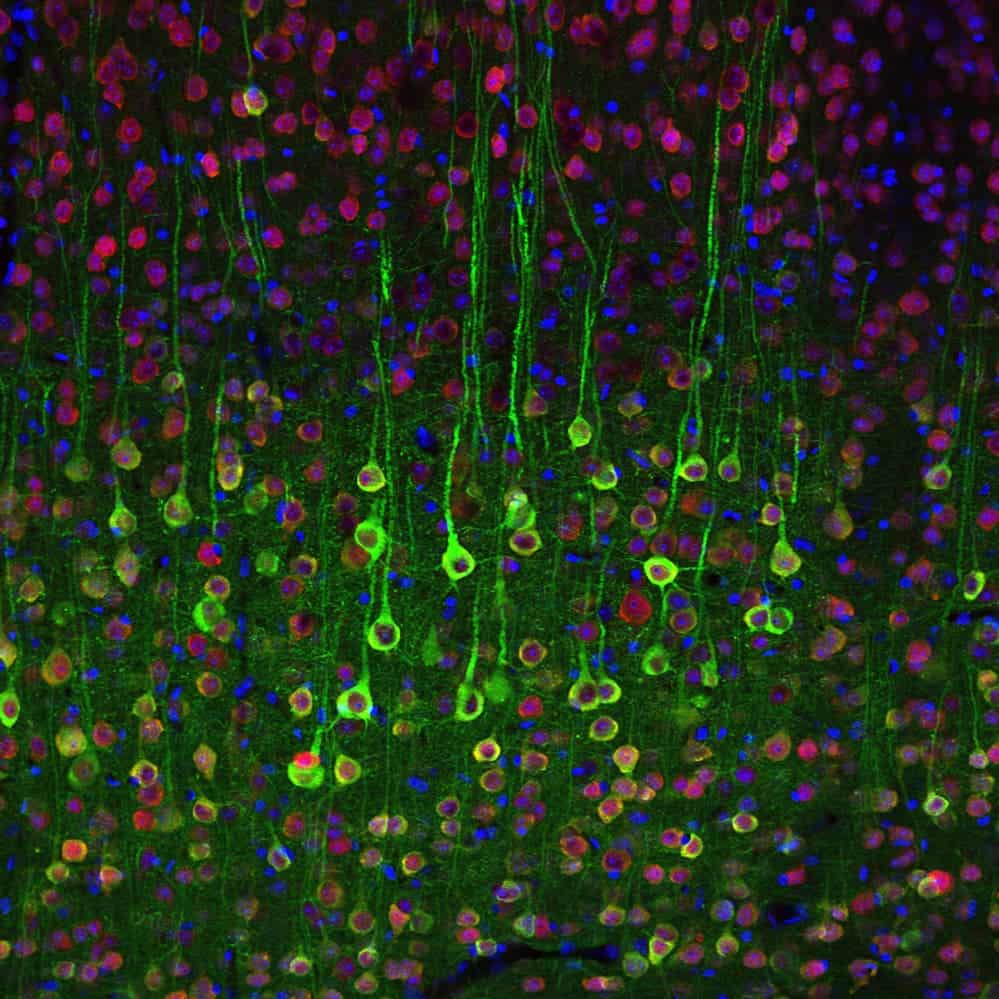
Using this technique, researchers managed to image a section of brain tissue 500 by 200 by 100 microns with a standard confocal microscope. Imaging such large samples would not be feasible with other super-resolution techniques, which require minutes to image a tissue slice only 1 micron thick. The resolution wasn’t diminished in any way either, which makes it even more spectacular.
“The exciting part is that this approach can acquire data at the same high speed per pixel as conventional microscopy, contrary to most other methods that beat the diffraction limit for microscopy, which can be 1,000 times slower per pixel,” says George Church, a professor of genetics at Harvard Medical School who was not part of the research team.
According to their study, this is the only way to truly analyze such samples.
“The other methods currently have better resolution, but are harder to use, or slower,” Tillberg says. “The benefits of our method are the ease of use and, more importantly, compatibility with large volumes, which is challenging with existing technologies.”
Source: MIT.






