New research at the University of Zurich (UZH) points the way towards a new type of anti-tumor treatment. This, they hope, will help protect patients from the side effects of cancer therapy.
Tumors are notoriously tricky to eliminate. The new approach, therefore, involves using our own bodies to produce therapeutic compounds at the tumor’s exact location. This should dramatically limit the negative side effects of traditional interventions because, unlike chemotherapy or radiotherapy, this approach does no harm to normal healthy cells. In fact, this approach could, potentially, also be used for targeted delivery of medicine against COVID-19 directly to the lungs.
Viral aides
It all revolves around a genetically-modified respiratory virus (an adenovirus, to be exact) which delivers genes encoding anti-cancer and signaling compounds directly into tumor cells. Here, they cause the cells to produce the very substances that destroy them, as well as chemical signals such as cytokines, which tells our immune system that the tumor is a target.
“We trick the tumor into eliminating itself through the production of anti-cancer agents by its own cells,” says postdoctoral fellow Sheena Smith, who led the development of the delivery approach.
“The therapeutic agents, such as therapeutic antibodies or signaling substances, mostly stay at the place in the body where they’re needed instead of spreading throughout the bloodstream where they can damage healthy organs and tissues” adds Andreas Plückthun, who led the research effort.
The team christened their new approach SHREAD: SHielded, REtargetted ADenovirus. It draws on the previous work of the same team, including ways to guide the virus to particular areas of the body, as well as methods to hide them from our immune system.
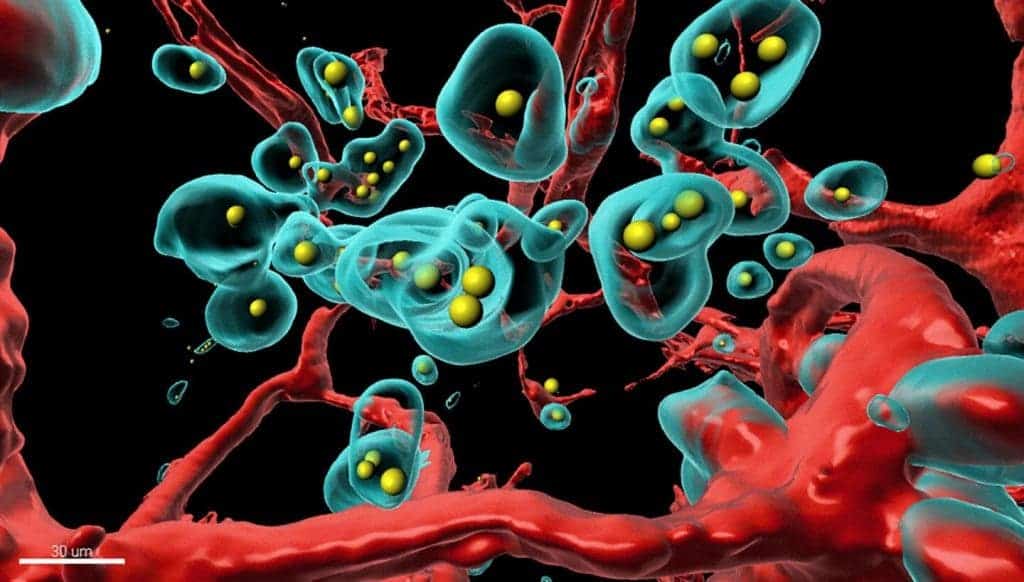
In order to test their approach, the authors used SHREAD to induce a breast tumor in the mammaries of a lab mouse to produce trastuzumab, a clinically approved breast cancer antibody. In a few days, levels of the antibody inside the tumor itself were higher than they could have been if they were injected directly. At the same time, they were significantly lower in the bloodstream and other tissues compared to what’s seen with direct injections — this helps reduce side effects.
The team used high-resolution 3D imaging methods and tissues rendered totally transparent to see how the antibody creates pores in blood vessels of the tumor and destroys tumor cells from the inside out.
One of the best parts of this approach is that it’s not limited only to cancer. The authors explained that by insulating healthy tissues from significant levels of an active substance, it also opens the door for other therapies. For example, it makes it possible to more easily use ‘biologics’, a family of protein-based drugs. If administered using a traditional injection, such biologics would be too toxic for use, they explain.
The team is currently working on applying SHREAD to anti-COVID-19 therapies.
“By delivering the SHREAD treatment to patients via an inhaled aerosol, our approach could allow targeted production of COVID antibody therapies in lung cells, where they are needed most,” Smith explains. “This would reduce costs, increase accessibility of COVID therapies and also improve vaccine delivery with the inhalation approach.”
The paper “The SHREAD gene therapy platform for paracrine delivery improves tumor localization and intratumoral effects of a clinical antibody” has been published in the journal PNAS.






