Developing safe, reliable, compact, and cost-effective hydrogen storage technologies is one of the most technically challenging barriers to the widespread use of hydrogen as a form of energy. Hydrogen is a great fuel, and if used in perfect conditions it can power numerous applications. However, with today’s tech a hydrogen powered car can’t even travel more than a few hundred miles without needing to refill, which makes it a highly frustrating alternative fuel. For the moment, that is.
Scientists have reported in a recently published paper, that they’ve successfully managed to synthesize a material capable of holding large amounts of hydrogen at ambient temperature and pressure. The study, authord by researchers from Peking University in Beijing, China, and Virginia Commonwealth University in Richmond, Virginia, US, has been published in a recent issue of Applied Physics Letters.
“Hydrogen storage is a big challenge for the hydrogen economy,” said Qiang Sun, who lead the research team. “Currently the research is behind schedule, but with the advancement of materials design and synthesis, reaching the target for future transportation applications [is becoming] more and more promising.”
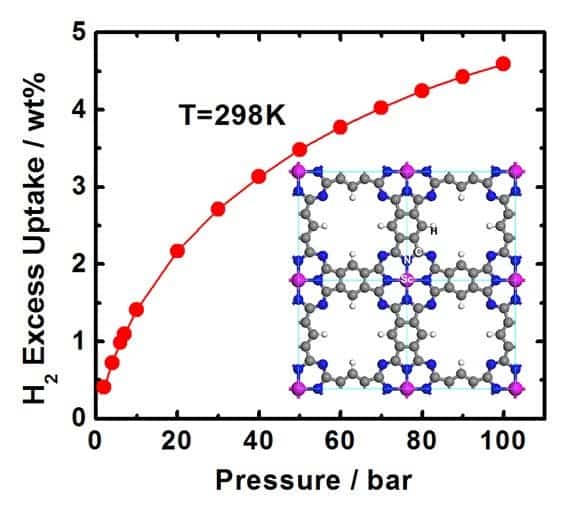
The US department of Energy had a goal for 2010 of storing hydrogen with a density of 4.3 wt. % (i.e., the hydrogen accounts for 4.3% of the total weight of the storage material) – this new material made out of metal-containing porous sheets is capable of storing hydrogen at a density of 4.6 wt. %. An impressing figure, by all means.
The biggest difficualty researchers had to face was that of balancing somehow the demanding characteristics of hydrogen in different conditions. You see, there have been previous attempts of developing materials capable of carrying high density hydrogen in the past, however stability has been an issue. For example, researchers once found that light metal hydrides can store hydrogen with a gravimetric density of 20 wt. %, however this made the storage material irreversible, and in consequence non-reusable. Who wants a storage tank that can only get filled once?
Other attempts have been made using materials such as carbon nanotubes and metal or covalent organic frameworks, which have been found to store hydrogen reversibly, however in these environments hydrogen adsorbs only at very low temperatures. The issue, researchers explain, lies in the bonding – in light metal hydrides, hydrogen is held in much stronger bonds than in carbon nanotubes, for instance. The key lies in finding an intermediate state.
“If the bonding is too strong, hydrogen can only be released at high temperature, while if the bonding is too weak, the storage is unstable at room temperature,” Sun explained. “So we require an intermediate bonding energy.”
For their study, the scientists investigated the performance of hydrogen storage in metal-containing porous sheets, made out of made of blue-green dye units called phthalocyanines (Pc) with regularly spaced iron (Fe) atoms or other metal atoms. The researchers systematically investigated 10 Pc-based porous sheets with transition metal atoms from scandium (Sc) through zinc (Zn), and found that porous Pc sheets with Sc atoms could store up to 4.6 wt. % hydrogen.
Hydrogen can be produced using abundant and diverse domestic energy resources, including fossil fuels, such as natural gas and coal; renewable energy resources, such as solar, wind, and biomass; and nuclear energy. This study might hold the key to overcoming critical challenges related to capacity, the uptake and release of hydrogen, management of heat during refueling, cost, and life cycle impacts.
Was this helpful?



