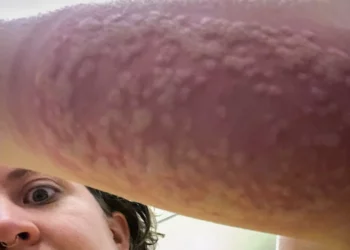
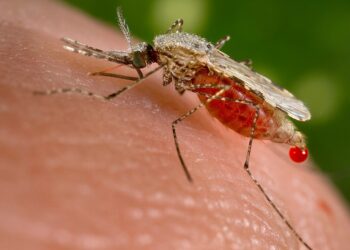
Despite the undeniable progress medical science has made in treating infectious diseases, malaria has always stuck like a thorn in humanity’s side. But the disease which infects 200 million people and kills half a million each year might have finally met its match: the 1st ever malaria vaccine. Emboldened by very promising clinical trials, the World Health Organization (WHO) announced the start of a pilot program that will see hundreds of thousands of children from Kenya, Ghana, and Malawi vaccinated against malaria.
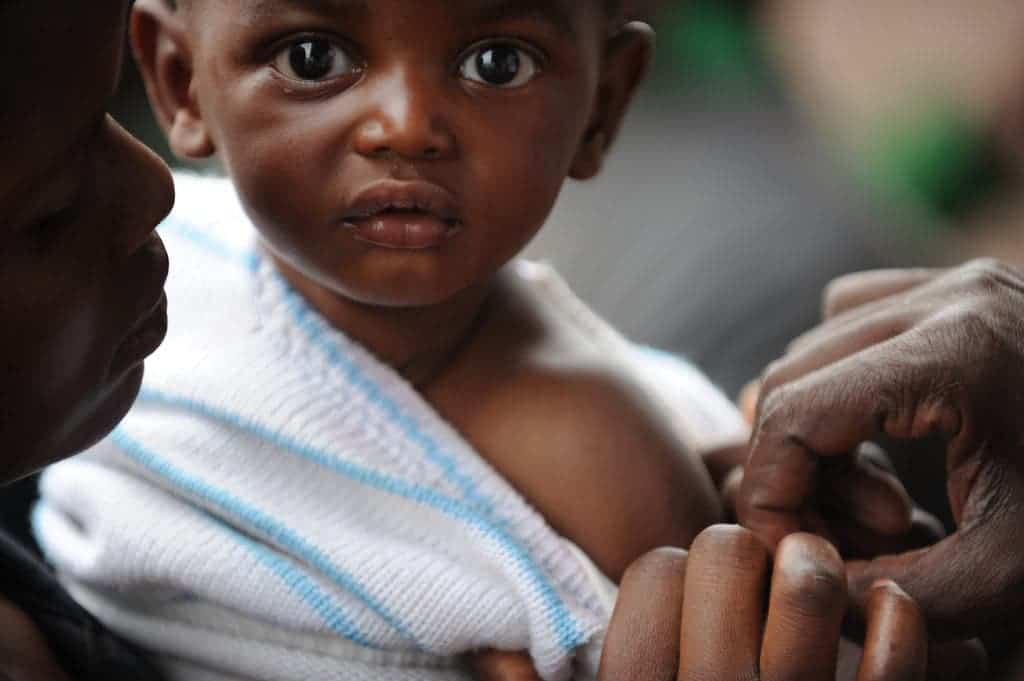
About 90 percent of cases of malaria occur in sub-Saharan Africa, and three African countries where the malaria vaccine will be piloted were not chosen at random. They’re some of the hardest hit countries by malaria but also have a solid track record of both monitoring and preventing malaria.
The first ever large-scale malaria vaccine trial will target children five to 17 months old. In each country, at least 120,000 children will be vaccinated to see whether the promising results from the clinical trials hold up to real-life conditions. The vaccine will be used alongside other preventative measures such as bed nets, insecticides, repellents and anti-malarial drugs, the WHO said.
The first malaria vaccine
The injectable vaccine RTS,S developed by the British pharmaceutical giant GlaxoSmithKline (GSK) in partnership with the PATH Malaria Vaccine Initiative took decades of work and billions to develop. It previously passed all of its clinical trials, including a phase three clinical trial between 2009 and 2014, and got approval for the large-scale pilot program in 2015.
The vaccine, also known as Mosquirix, does not promise full protection against the mosquito-borne disease, but it is the most successful scientifically-vetted malaria vaccine at our disposal. Trials so far involving 15,000 participants from seven countries suggest RTS,S reduces the frequency of malaria episodes by 40 percent. Given the scale of malaria infection on a yearly basis, this means the vaccine could save hundreds of thousands of lives.
“It’s an efficacy rate which is quite low, but given the amount of affected people, the impact will be huge,” said Mary Hamel, who is coordinating the vaccine’s implementation programme.
“There will be other vaccines and they’ll be more efficient, but in the meantime, this will have a significant influence.”
Another malaria vaccine is the Sanaria® PfSPZ-CVac, which was first tested in 2013. A phase-1 clinical trial which involved 67 healthy adult test persons none of whom had previously had malaria found the vaccine was 100 percent effective when assessed at 10 weeks after the last dose of vaccine. Another clinical trial, this time conducted in a malaria hotspot in Mali, Africa, found the vaccine was 48 percent effective at preventing infection.
Hopefully, all three African countries will rise up to the challenge. To be effective, the vaccine must be delivered in four successive doses given on a strict timetable, which isn’t at all easy to organize.
If the vaccine is found to be effective, it will likely be adopted on an even greater scale across Africa, but also Latin America and the Middle East which also report quite a lot of malaria cases. Between 2000 and 2015, preemptive measures have been effective at cutting 62 percent of all malaria-related deaths. The WHO hopes to eradicate malaria by 2040. With a bit of luck and a lot of hard work, this pilot program might be remembered a couple of decades from now as a huge milestone in our quest to say goodbye to malaria once and for all.