A vaccine against malaria has proven to be 77% effective in trials in Africa and could be a major breakthrough against the disease, according to its developers from the Jenner Institute at the University of Oxford.
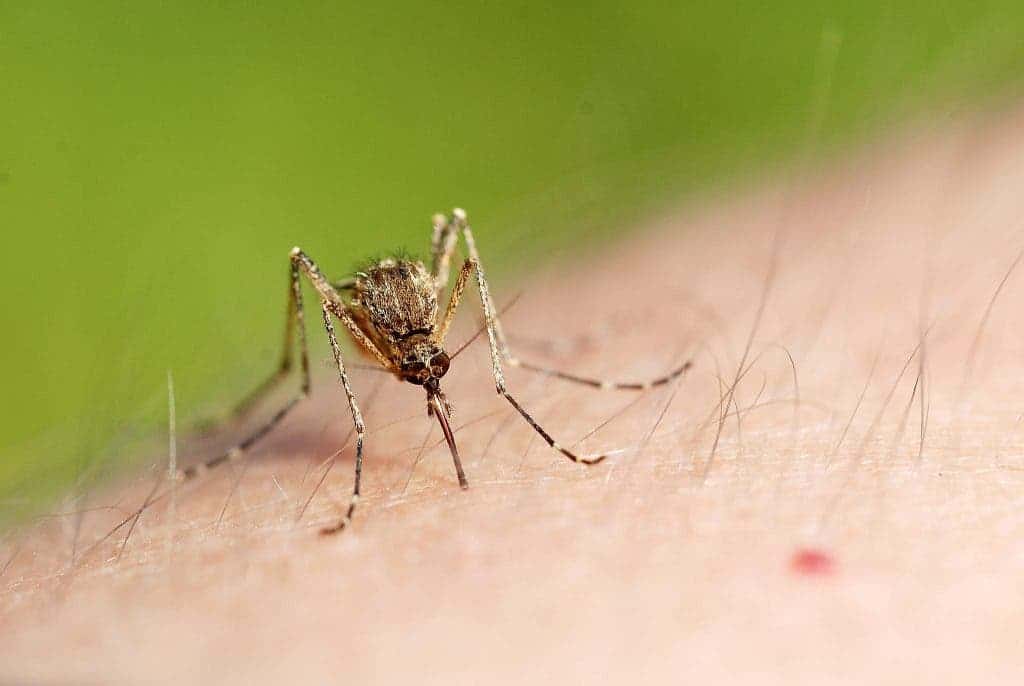
Malaria kills over 400,000 people every year, mostly small children in sub-Saharan Africa; one child dies from the disease every two minutes, according to the World Health Organization (WHO). Although many vaccines have been trialed over the years, none has been sufficiently successful.
A parasitic disease, malaria is transmitted through the bite of female Anopheles mosquitoes. It is both preventable and treatable. The African region was home to 94% of all malaria cases and deaths in 2019, according to the WHO. In recent years, countries have made progress using new tools such as insecticide-treated mosquito nets.
The Oxford vaccine, known as R21, is the first one to meet the WHO goal of 75% efficacy against the mosquito-borne parasite disease. The previously developed Mosquirix vaccine, now being piloted by the WHO in four countries in Africa, was partially effective, preventing 39% of malaria cases among small children in Africa over four years. A 75% efficacy could be a game changer.
“These are very exciting results showing unprecedented efficacy levels from a vaccine that has been well-tolerated in our trial programme,” Halidou Tinto, the trial’s principal investigator, said in a statement. “We look forward to the upcoming Phase III trial to demonstrate large-scale safety and efficacy data for a vaccine that is greatly needed in this region.”
Trialed in 450 children between the ages of five and 17 months in Burkina Faso, the vaccine was found to be safe and showed “high-level efficacy” over 12 months of follow-up. The children were divided into three groups and the higher dose one was 77% less likely to get the disease, the researchers reported in a pre-print study in The Lancet.
There were “no serious adverse events related to the vaccine,” the researchers wrote in the study. Now, together with their commercial partners the Serum Institute of India and drugmaker Novavax, they are recruiting for a Phase III trial to assess the safety and efficacy of the vaccine in 4,800 children, aged from five to 36 months.
Adrian Hill, director of the Jenner Institute, where the Oxford/AstraZeneca Covid vaccine was recently invented, told The Guardian that the vaccine has the potential to cut the death toll from malaria significantly. Even going from the 400,000 annual deaths to “tens of thousands” in the next five years, if the vaccine proves successful.
Hill said the institute will likely apply for emergency approval for the malaria vaccine just as it did for the COVID-19 jab. “Malaria kills a lot more people than Covid in Africa, so you should think about emergency-use authorization for a malaria vaccine,” he said. The institute will first ask regulatory bodies for a scientific opinion on the vaccine.
The researchers said the vaccine will be manufactured on a large scale and low cost. They have already done a deal with the Serum Institute of India, which is involved in manufacturing the Oxford/AstraZeneca Covid-19 vaccine. The Serum Institute promised to deliver 200 million doses a year of the malaria vaccine if it’s licensed.
Cyrus Poonawalla and Mr Adar Poonawalla, Chairman and CEO of the Serum Institute of India said in a statement: “We are highly excited to see these results on a safe and highly effective malaria vaccine. Serum Institute is committed to global disease burden reduction and disease elimination strategies by providing high volume, affordable vaccines.”
Was this helpful?



