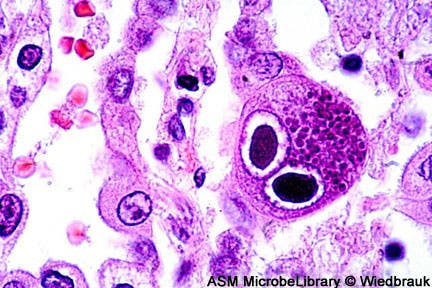
A virus that most of us carry, yet which is remarkably obscure both to the immune system and the general public in terms of awareness, is responsible for a number of health hazards in the human body. In time, it tires the immune system which is forced to seek and fight it for a life time, exposing the body to other health hazards, and is the number one infectious cause of congenital birth defects. Researchers at Cardiff University and the La Jolla Institute, California, have now made great strides forward in killing the virus after they identified the cellular mechanism it uses to infect its host body, thus providing key information that one day might allow an effective vaccine to be developed.
One child in 750 is born with a congenital defect like hearing or brain damage as a result of cytomegalovirus (CMV) infection – much more than other well-known congenital problems, such as Down’s syndrome or fetal alcohol syndrome. Despite this, few people are aware of its existence or of the fact they carry it.
What makes it so dangerous is its stealthy nature which makes it very difficult for the immune system to identify it. To top things over, the virus belongs to the herpes family of viruses that cause cold sores, chicken pox and other maladies, and like its brethren it never goes away. Also, the constant struggle and stress the immune system is subjected to as it tries to control the virus tires and weakens it in the process, exposing it to other maladies.
“We have identified a novel trick that this virus uses to hide from immune detection,” says La Jolla Institute scientist Chris Benedict, Ph.D., a CMV expert. “By uncovering this mechanism, we’ve provided an important piece of the CMV puzzle that could enable vaccine counter strategies that flush out and eliminate virus-infected cells.”
A well hidden pest
What the researchers found was that specific CMV gene (called UL141) blocks the ability of two key immune pathways to kill CMV-infected cells. These are TRAIL “death receptor” 1 and 2, first discovered some 15 years ago that normally work to kill infectious cells. The TRAIL death receptors have been the subject of study for oncologists for many years, interested in targeting them for anti-tumor therapies.
By identifying the UL141 gene as an inhibitor of TRAIL’s ability to carry out its killing function, the researchers hope this new found information will play a pivotal role in developing an effective vaccine against the CMV virus.
“This finding puts on the table the importance of TRAIL signaling in host (our body’s) defense and how the virus works to block these efforts,” says Ed Mocarski, Ph.D., a scientist at the Emory University School of Medicine’s Vaccine Center, whose research focuses on new ways to combat CMV. “This knowledge could set the stage for developing ways to boost the adaptive immune response which could ultimately aid in developing an effective vaccine.”
The findings were reported in the journal Cell Host & Microbe.