The Thomson model, most commonly called the “Plum Pudding” model, was an early attempt to explain what the structure of the atom was like. While the attempt did not really describe the atomic structure accurately, it got a few things right, which set the stage for further developments in atomic research. The plum pudding model was an important departure from earlier atomic models that deserves its place in history.
The JJ Thompson model
In 1904, British physicist J. J. Thomson proposed his idea of what the atom looked like. It was an unusual, but plausible construction that was well-received in the scientific community. Whereas early atom models said that atoms were indivisible and had no internal structure, Thomson argued that the atom must have a structure consisting of some things that are positively charged and other things that are negatively charged.
A few years earlier, Thomson had discovered the electron — a small, subatomic, negatively charged particle. Thomson was trying to reconcile two of the known properties of the atom: that electrons have a negative charge, and the overall atom is neutral.
If electrons are negative and the atom is neutral, then something must be positive. So Thomson came up with a distribution that made sense at the time — in quaint British fashion, he called it the Plum Pudding Model.
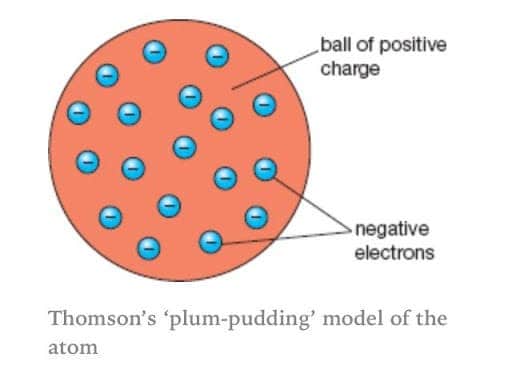
The Plum Pudding Model has electrons surrounded by a volume of positive charge in which negative-charge electrons are embedded. It was relatively short-lived: in 1911, Ernest Rutherford published results showing a small nucleus of positive charge, instead of a large mass around the electrons.
So just to clarify: we now know that the plum pudding model is inaccurate. But it’s useful to learn about it, see how it was developed, and how scientists realized that something else was happening.
An atomic model
The idea that matter is made of atoms goes back a long time, but for two thousand years, it fell out of the spotlight.
The Greek philosopher Democritus, born around the year 460 BC, promoted what was regarded by many as a crazy idea: everything we see around us is made of small, individual constituents. He called these constituents “atomos”, or atoms, as we’d call them in English.
Some of the ideas Democritus had turned out to be remarkably correct, showing just how good the intuitive understanding of the Greek philosopher was. He believed that atoms are invisible to the human eye, but they do have a geometry and an internal structure. They are always in motion, are separated by void, and individual atoms are indestructible — we now know atoms are not indestructible, but much of what Democritus said is on point.
Although some philosophers still considered it, the atomic idea of Democritus was largely abandoned for centuries, particularly as it was disregarded by his followers Plato and Aristotle. Plato, who believed that all matter is made of the four elements (earth, fire, wind, and water), reportedly hated Democritus so much that he wished all his books were burned.
Nevertheless, Democritus is considered by some the “father of modern science“, his approach being closer to science than to philosophy, as was the case of Plato, who is widely cherished to this day.
But while Democritus was close to the truth, his idea was largely ignored for centuries because it was not possible to prove it. However, that changed dramatically in the 20th century, as new scientific discoveries were being made. At some point, philosopher and polymath Bertrand Russel wrote that this atomic theory “was remarkably like that of modern science, and avoided most of the faults to which Greek speculation was prone.”
The atomic theory had once again come to attention.
The Thomson model
Near the end of the 18th century, English researcher John Dalton was struggling to explain some experimental results. Dalton, who also studied color blindness (hence the term “daltonism”), found that no matter how you combine chemical elements, the total mass in the reaction remains the same. He and other chemists also noted that water absorbs different gases in different proportions — for example, water absorbs carbon dioxide far better than it absorbs nitrogen.
He proposed that each chemical element is composed of atoms of a single, unique type. These atoms cannot be altered or destroyed by chemical means, but they can combine to form more complex structures — strikingly similar to what Democritus had proposed.
This was the dominant school of thought for about one century — until one J. J. Thomson came along.

Plum pudding model
Rather ironically, the ‘Plum Pudding Model’ is actually a misnomer. It uses an archaic meaning of the British ‘plum pudding’, which is actually a cake made with raisins (in pre-Victorian times, ‘plum’ was a term used for raisins). But this old-school cake would take yet another meaning.
By the early 1900s, famous researchers such as Amadeo Avogadro, Robert Brown, and even Albert Einstein had explored the flaws of the Dalton model — but couldn’t quite come up with a better alternative. Thomson himself continued to explore the components of the atom and was actively looking for answers.

Atoms were still thought to be the smallest possible division of matter until Thomson’s experiments in 1897. Through experimentation, Thomson found that a cathode ray can be deflected by an electric field. This means that instead of being light, this cathode ray had to be something else, which Thomson correctly deduced to be a flow of electrons. In other words, he had found one of the building blocks of atoms: a negatively charged electron. It was a particle unlike any previously known, and Thomson took this time before he could prove his discovery beyond the shadow of a doubt.
He even measured the mass-to-charge ratio of electrons, finding that they are 1800 times smaller than hydrogen, the smallest atom. There was no doubt about it: atoms were divisible, and these electrons were negative in charge. Which means something must also be positive in atoms.

If you want to bake a plum cake, you must first invent the universe
Atoms are neutral in electric charge. Sure, there are ions that are positively or negatively charged, but technically speaking, atoms are neutral. So if electrons are negative, they must have a positive counterpart.
Since Thomson found electrons to be so small, whatever that positive thing must be should be relatively big. So he considered three scenarios:
- Each negatively charged electron has a positively charged particle that follows it everywhere;
- Electrons orbit a region of positive charge with the same magnitude as the total charge of all electrons;
- The negative electrons occupy a space that is uniformly and positively charged.
He selected the latter as the most likely of the three. He submitted his ideas to the 1904 edition of the Philosophical Magazine, where Thomson wrote:
… the atoms of the elements consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification, …
This theory was embraced by physicists, who started devising experiments to learn even more about atoms, based on this configuration.
Ironically, it was these exact experiments that ended up disproving the Plum Pudding model.
Disproving the cake, and raisin’ a new theory
Funny enough, Thomson’s model was disproved by one of his students, Ernest Rutherford. This just goes to show how influential Thomson’s work and lab were at the time.
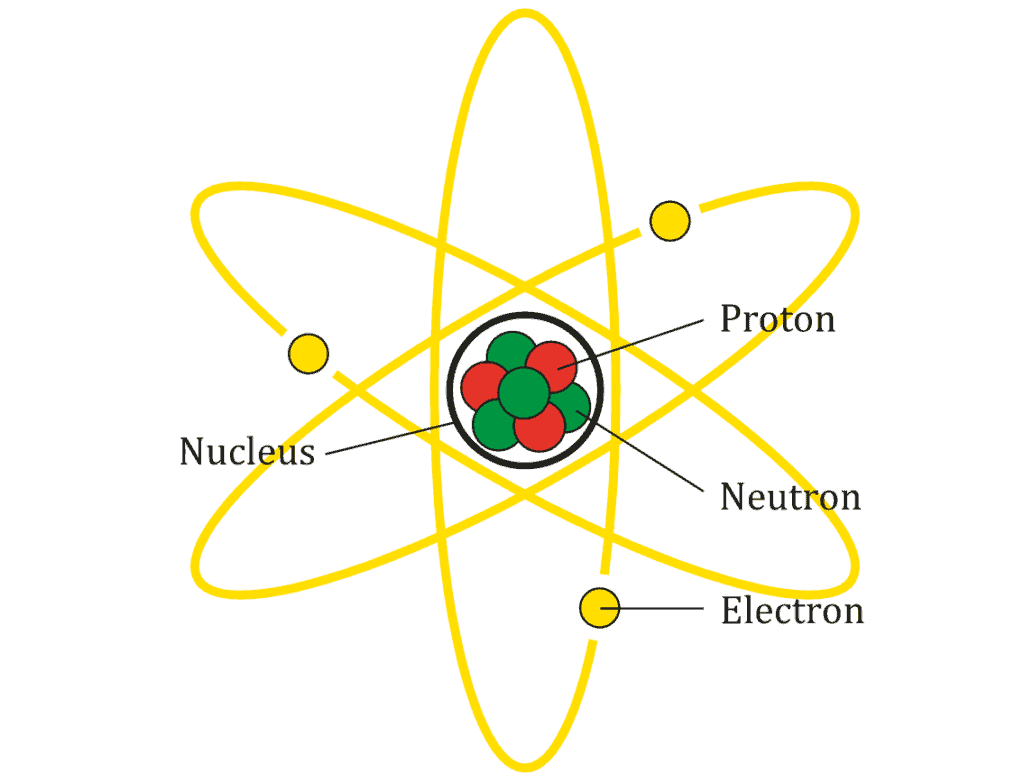
Rutherford’s experiments showed that the positive charge is concentrated towards the center of the atom, in what seemed to be an atomic nucleus. This would mean that the negative charge (the electrons) would be concentrated outside of the atom.
Rutherford immediately suspected a planetary model of the atom, where the nucleus is like a star and the electrons orbit around it like planets. But there was a problem, and a big one at that: it contradicted classical mechanics.
In Rutherford’s envisioned model, the electron would release electromagnetic radiation while orbiting a nucleus. This means it would lose energy in the process, spiraling closer to the nucleus, and collapsing on the atom in no time.
This model is a disaster because it would suggest that all atoms are unstable, which is clearly not the case. So the planetary atom model is tempting, but it lacks something fundamental, and was initially disregarded.
It was only when Niels Bohr’s explanation came along that this model could find its ground.
According to Both, the electron is able to revolve in certain stable orbits around the nucleus without radiating any energy, contrary to what classical electromagnetism suggests. Rutherford and Bohr presented their model together, and although our understanding of the atom has changed several times and we’ve learned far more about atomic and subatomic particles, this model is still widely used, at least in non-academic circles.

Bohr’s model was not perfect. Fast forward to modern times and we have a substantially better understanding of atoms, but we’re still trying to reconcile various aspects of atom structure (particularly quantum physics).
Why bother learning about the Plum Pudding model

It’s easy to disregard the Plum Pudding model as flawed or imperfect and never look at it again. But there’s a reason why physics classes still feature this model, and it’s not just for historical reference.
The idea is that if we want to truly learn something, and not just memorize it, it helps to build a process. If we go through the stages of how physicists first learned about atoms, what theories they had, and how these theories were proven or disproven, we gain a much better understanding. That’s why we learn about the Plum Pudding model, because there can be value even in discredited ideas.
Rest assured: as science and technology progress, no doubt some of the models we use today will turn out to be flawed, and people will still learn about them.
We’re still learning new things about the atoms’ constituents and their structure, and we’ll be learning more for the foreseeable future. Science is rarely about finding an ultimate, finite truth — instead, science is about adding more and more layers of understanding and building approximate models. Thomson’s model was one of these approximations: it was far from perfect, in fact, it was thoroughly disproven — and yet, it played an important role, as it paved the way for more, better discoveries to be made.
This is what science is all about.






