Though Earth’s iron-rich core is subjected to a dizzying temperature in excess of 5,427° C ( 9,800° F), it stays solid. This has always been a mystery for geologists and geophysicists but novel research might have finally settled the debate.
According to Swedish physicists from the KTH Royal Institute of Technology, the iron inside the core stays crystallized because “it exhibits a pattern of atomic diffusion.”
The Earth’s core, the innermost part of our planet, is completely out of our direct reach. The deepest hole we’ve ever dug is about 12 kilometers, whereas the core lies at about 2,900 kilometers deep, and the inner core is at starts at 5150 km.
But we can know some things about it by looking at seismic waves.
When an earthquake happens, it sends out seismic waves that get picked up at seismographs around the planet. Sometimes, these waves propagate through the core as well, and based on how they propagate through the core, we can infer some things about it.
For instance, we know with certainty that the inner core is solid — which sounds completely surprising, given how hot it is.
The full picture
Much of what we know about the Earth’s inner structure is owed to some clever people who realized a long time ago that they could use acoustic physics to infer properties of objects, even when they are obscured by thousands of kilometers of material. In a fluid, such as a gas or liquid, you can get shear waves when a pressure wave hits a surface.
While a pressure wave’s wobble is always in the same direction, a shear wave is like a wave on a string (it’s at a right angle with the wave’s direction of propagation). When waves created by earthquakes hit the liquid outer core and then travel through the inner core, scientists record an extra wave going off at right angles which can only be explained by a shear wave.
The only explanation is that core must be solid.
As mentioned, seismic waves, which are essentially waves of energy that travel through the Earth’s interior, provide valuable information about the structure of the planet, and researchers can tell whether these waves are passing through a solid or liquid medium. In the case of the inner core, there’s no doubt about it: the inner core is solid.
So how is it solid when it’s so hot?
Well, whether something remains solid or melts depends firstly on temperature — but it also depends on the chemistry (what the object is made of) and pressure. Higher pressure raises the melting point as well.
This solid inner core, which is nearly the size of the moon, is mostly made of crystallized iron.
The Earth’s core formed early in the planet’s history, with heavier elements sinking towards the center, when the entire planet was just forming. The core is kept hot by the decay of radioactive elements within the Earth, as well as residual heat from the planet’s formation.
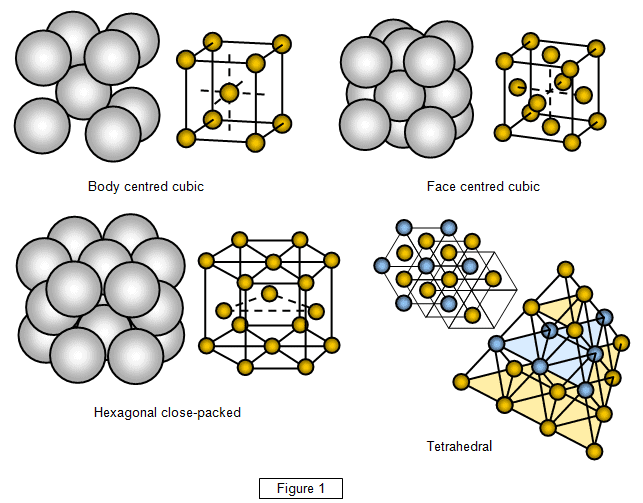
Being a metal, iron’s atomic crystal structure is packed at room temperature and pressure in a body-centered cubic (BCC) phase — an architecture with eight corner points and a center point. At extremely high pressures, the crystalline structure transforms into 12-point hexagonal forms known as a close-packed (HCP) phase.
Seeing how Earth’s core is subjected to pressures 3.5 million times higher than atmospheric pressure, never mind temperatures more than 5,000 degrees K higher, it would be sensible to assume an HCP, liquid phase. But the evidence says otherwise.
Using a supercomputer, Anatoly Belonoshko, a researcher in the Department of Physics at KTH, and colleagues, explained for the first time what was going on.
In short, characteristics of BCC iron once thought to render it unstable at high pressure actually cause the opposite effect — it makes the BCC crystalline phase even more stable.
“Under conditions in Earth’s core, BCC iron exhibits a pattern of atomic diffusion never before observed,” Belonoshko says.
“It appears that the experimental data confirming the stability of BCC iron in the Core were in front of us – we just did not know what that really meant,” he added, alluding to observations collected three years ago at Livermore Lawrence National Laboratory in California.
At high pressure but low temperature, the BCC phase is unstable as the crystalline planes slide out of the cubic structure. However, analysis of the computational samples suggests at that at high temperatures, these structures stabilize much like cards in a deck.
“The sliding of these planes is a bit like shuffling a deck of cards,” says Belonoshko. “Even though the cards are put in different positions, the deck is still a deck. Likewise, the BCC iron retains its cubic structure. The BCC phase goes by the motto: ‘What does not kill me makes me stronger.’ The instability kills the BCC phase at low temperature, but makes the BCC phase stable at high temperature.”
The data also revealed the most accurate composition of the inner core to date: 96 percent iron, the rest nickel and possibly light elements.
Belonoshko also said that the shuffling or diffusion of atoms can also explain another inner core mystery: why seismic waves travel faster between the Earth’s poles than through the equator. In an anisotropic material, its properties change with the direction of the object. Like a grain of wood, Earth’s inner core also has a texture that changes with direction, being anisotropic.
“The unique features of the Fe BCC phase, such as high-temperature self-diffusion even in a pure solid iron, might be responsible for the formation of large-scale anisotropic structures needed to explain the Earth inner core anisotropy,” he says. “The diffusion allows easy texturing of iron in response to any stress.”
So the outer core is the only liquid layer of the Earth (contrary to popular belief, the mantle is also solid, though it can move a bit like a liquid in geologic time). The inner core is solid — and now we truly understand why.
Findings appeared in the journal Nature Geosciences. Check the video below for a visual explanation of what’s going on inside our planet’s core.