Scientists from the Scripps Research Institute (TSRI) and Janssen Pharmaceutical Companies of Johnson & Johnson (Janssen) have discovered a way to give antibodies the ability to fight a wide range of influenza subtypes. Their work has great potential to one day eliminate the need for repeated seasonal flu shots.
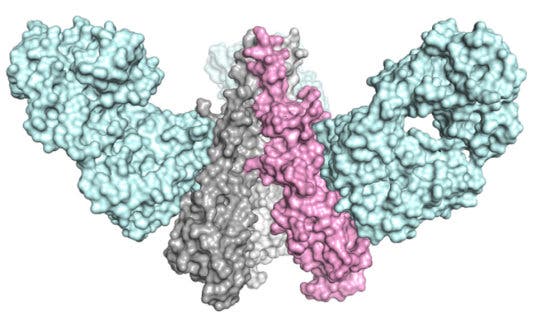
Image credits to The Scripps Research Institute
The research was published online ahead of print on August 24 by the journal Science.
We need better flu vaccines
Every fall, millions of people roll up their sleeves for a flu vaccine (there’s no way a vaccine will give you the flu, find out why here), hoping to give their immune system a leg up on influenza. But the flu virus has thousands of strains that mutate and evolve across seasons, and the vaccine can’t guard against all of them.
Seasonal flu typically causes more than 200,000 hospitalizations and 36,000 deaths every year in the United States, according to the U.S. Centers for Disease Control and Prevention. While yearly flu shots provide some protection, when subtypes not covered by the vaccine do emerge, they can cause a lot of damage. This was evident in the 2009 spread of the H1N1 swine flu subtype that killed an estimated 151,700 to 575,400 people worldwide.
The team hopes to change all that by allowing antibodies to attack a wide range of flu strains. Several studies performed in the last decade by TSRI, Janssen and other institutions have shown that some patients are capable of making powerful antibodies that can fight many subtypes of influenza at once by targeting a site on the influenza virus that does not mutate rapidly. Unfortunately, these “broadly neutralizing antibodies,” or bnAbs, are rare.
Researchers searched for and found a specific protein on the surface of the influenza virus, called hemagglutinin (HA). The protein is present on all subtypes of influenza, and it underpins the viral mechanism for cell infestation. More to the point, the longer, “stem” region of the HA protein, that works to connect the virus to the cell it attacks, plays such a crucial role for influenza viruses that mutations at that site are unlikely to be successful and passed on. Janssen and TSRI tried to make a vaccine that elicits broadly neutralizing antibodies to specifically target the HA protein.
“If the body can make an immune response against the HA stem, it’s difficult for the virus to escape,” Wilson explained.
One vaccine to rule them all
The effort represents the first time scientists have been able to cut off the variable head region of HA, designing features able to stabilize the conformation of the original protein, and at the same time faithfully mimicking the key broadly neutralizing site.
In order to create antibodies that can tie to the HA stem, the researchers looked at influenza’s structure, specifically the universal recognition site (the area where the antibody ties to the virus) of the antibody CR9114 in the HA stem.
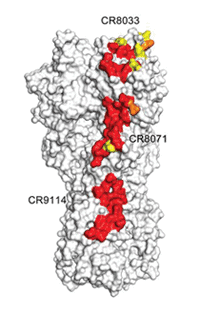
Image via als.lbl.gov
The vaccine candidate was designed, produced and tested by a team of scientists led by Jaap Goudsmit, head of the Janssen Prevention Center, the paper’s first author Antonietta Impagliazzo (responsible for the design) and co-senior author Katarina Radošević. The ultimate goal was to use this synthetic version of the HA stem in a vaccine to teach the body to make powerful antibodies against influenza virus, priming it to fight off a variety of flu strains.
The scientists then studied the response of rodent and nonhuman primate models given one of several candidate immunogens. They found that animals given one especially stable immunogen produced antibodies that could bind with HAs in many influenza subtypes, even neutralizing H5N1 viruses (“bird” flu).
“This was the proof of principle,” said Wilson. “These tests showed that antibodies elicited against one influenza subtype could protect against a different subtype.”
Scientists studied the structure of the immunogen at every point in the process. Using imaging techniques such as electron microscopy (led by TSRI Associate Professor Andrew Ward and postdoctoral fellow Ryan Hoffman) and x-ray crystallography (led by Wilson and TSRI Staff Scientist Xueyong Zhu), the team showed that the most promising candidate immunogen mimicked the HA stem and that antibodies could bind with the immunogen just as they would with a real virus.
With proof that an immunogen can elicit antibodies against the stem region, Wilson said the next step in this research is to see if the immunogen can do the same in humans.
“While there is more work to be done, the ultimate goal, of course, would be to create a life-long vaccine,” Wilson said.
“This study shows that we’re moving in the right direction for a universal flu vaccine,” said Ian Wilson, Hansen Professor of Structural Biology and chair of the Department of Integrative Structural and Computational Biology at TSRI.
For more influenza vaccine articles click here.






