What nature seems to perform effortlessly with photosynthesis has proven to be an immense hurdle for scientists trying to mimic it with so called artificial leafs. In recent years, important breakthroughs have been made with this scope in mind, yet the artificial leaf is so inefficient at the moment that it’s absolutely not worth pursuing on a large scale. Researchers at Arizona State University report considerable advances toward perfecting a functional artificial leaf.
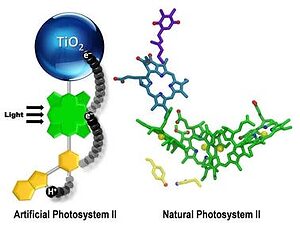
- An artificial photosynthetic reaction center containing a bioinspired electron relay (yellow) mimics some aspects of photosynthesis. Photo: ASU
Plants are extremely efficient at taking carbon dioxide from the air and converting it into biomass using light as an energy source. What scientists are particularly interested in, however, is not in mitigating carbon but using an artificial photosynthesis-like process to break down water into its constituting molecules: hydrogen and oxygen. Hydrogen is one of the most powerful and easy to use fuels at our disposal, the problem is producing it. This is the whole basis of the “hydrogen economy” – a platform that envisions a world where energy needs are met by hydrogen fuel, cheaply and cleanly obtained. Society requires a renewable source of fuel that is widely distributed, abundant, inexpensive and environmentally clean.
An artificial leaf
The thing is, as scientists first understood some decades ago, mimicking photosynthesis is a whole lot harder than it seems. The researchers took a closer look at how nature had overcome a related problem in the part of the photosynthetic process where water is oxidized to yield oxygen.
“Initially, our artificial leaf did not work very well, and our diagnostic studies on why indicated that a step where a fast chemical reaction had to interact with a slow chemical reaction was not efficient,” said ASU chemistry professor Thomas Moore. “The fast one is the step where light energy is converted to chemical energy, and the slow one is the step where the chemical energy is used to convert water into its elements viz. hydrogen and oxygen.”
“We looked in detail and found that nature had used an intermediate step,” said Moore. “This intermediate step involved a relay for electrons in which one half of the relay interacted with the fast step in an optimal way to satisfy it, and the other half of the relay then had time to do the slow step of water oxidation in an efficient way.”
With this new found knowledge, the researchers constructed an artificial relay based on the natural counterpart. They then designed an artificial relay based on the natural one and were rewarded with a major improvement. X-ray crystallography and optical and magnetic resonance spectroscopy techniques were used to determine and characterize the interactions occurring between electrons and protons participating in the relay. There’s a rather solid theory on proton coupled electron transfer mechanism; with this theoretical work to guide them and the experimental data at hand, the Arizona scientists identified a unique structural feature of the relay. This was an unusually short bond between a hydrogen atom and a nitrogen atom that facilitates the correct working of the relay.
They also found subtle magnetic features of the electronic structure of the artificial relay that mirrored those found in the natural system.
Not only has the artificial system been improved, but the team understands better how the natural system works. This will be important as scientists develop the artificial leaf approach to sustainably harnessing the solar energy needed to provide the food, fuel and fiber that human needs are increasingly demanding.
source: ASU






