Water is the essence of all life as we know it, but physically and chemically, water still holds plenty of mysteries.
Water molecules exist in two forms — different, but with almost identical physical parameters. Researchers refer to these two forms as ortho-water and para-water. Now, a new study has mapped their different properties.

Since middle school, we’ve learned the basics of water: it’s made from two hydrogen atoms and one oxygen atom, it’s a liquid, almost ubiquitous on Earth, and it is essential for life as we know it. Water also has a few peculiarities. For instance, water is most dense at 4 °C and is least compressible at 46 °C — two temperatures that don’t seem to have any particular meaning. But water has another quirk, researchers found: it comes in two shapes (isomers).
Two types of water
Water is not the easiest thing to study. Several different methods have been used to investigate water, but stunningly, different studies sometimes produced different results. This was not only confusing but also caused academic disputes. But as it turns out, a part of that could be explained by the fact that there’s more than just one type of water.
The two types of water are isomers — compounds that contain the exact same number of atoms (and have the same chemical formula) but differ in the way their atoms are arranged.
Pure water consists of two hydrogen atoms and one oxygen atom. The difference between the two water isomers lies in the relative orientation of the nuclear spins of the two hydrogen atoms. The nuclear spin, in this case, is the intrinsic form of angular momentum carried by the nucleus. Depending on how the spins are aligned (in the same or opposite directions), we end up with ortho- or para-water.
“Because it contains two hydrogen atoms, water has two different nuclear spin states: ortho- and para-water. In ortho-water the hydrogen nuclear spins are in the same direction, while in para-water they are opposite to one another,” explains study lead author Shaoqing Du, co-author of a recent study on the two different types of water. “Understanding the transition between these two types of water is an important area of research.”
In one study, a team headed by Professor Stefan Willitsch from the University of Basel in Switzerland investigated how the two forms of water differ in terms of their chemical reactivity — in other words, how the two behave differently during chemical reactions.
Since the physical properties of the water isomers are extremely similar it’s a very difficult thing to measure. However, by using an innovative process developed by Professor Jochen Küpper from the Hamburg Center for Free-Electron Laser Science, researchers were able to carry out controlled reactions that allowed them to measure these slightly different properties. They found that para-water reacts about 25% faster than ortho-water — probably because the spin influences the rotation of the water molecules — and this affects how quickly they interact with other molecules. As a result, para-water is able to attract its reaction partner more strongly than the ortho-form, which leads to increased chemical reactivity. The experimental results were also confirmed by computer models.
In another study, researchers at University of Tokyo, Kyoto University, and Tohoku University used a “molecular cage” to study water.
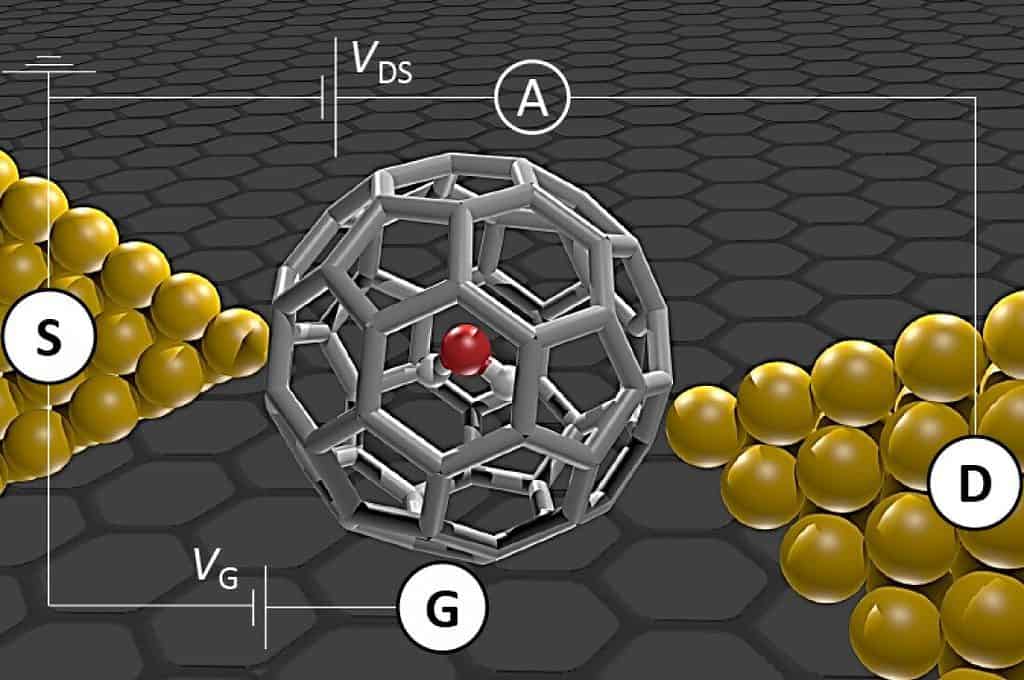
A C60 cage is a soccer ball-shaped molecule made of carbon atoms, which is an excellent way for isolating water and studying it. They found that within a minute, the single water molecule transitioned between the two nuclear isomers (ortho- and para-water) within one minute.
It’s not clear exactly why this happens, but it probably has to do with this fundamental property of water that we’re still not understanding perfectly.
“Our findings make an important contribution to the understanding of ortho-para fluctuation in water molecules,” says study corresponding author Kazuhiko Hirakawa. Because water plays such an important role in chemistry and biology, and even in understanding our universe, we expect our findings to have a wide-ranging impact.
While isolated water molecules can stay in their ortho or para form, in real-life conditions, there are constant molecular collisions between molecules that quickly mix the two forms up.
So for our day-to-day lives, this doesn’t make any difference at all — we can’t really taste ortho- or para- water. In fact, the researchers had to get the molecules at extremely low temperatures close to the absolute zero point (about –273°C) to even observe these different properties. Willitsch explains:
“The better one can control the states of the molecules involved in a chemical reaction, the better the underlying mechanisms and dynamics of a reaction can be investigated and understood.”
So at the end of the day, it doesn’t change much unless you’re a chemist or a physicist, but we can add another entry to the long list of water’s peculiarities.
Journal Reference: Ardita Kilaj, Hong Gao, Daniel Rösch, Uxia Rivero, Jochen Küpper, Stefan Willitsch Observation of different reactivities of para- and ortho-water towards trapped diazenylium ions Nature Communications (2018), doi: 10.1038/s41467-018-04483-3
Shaoqing Du et al, Inelastic Electron Transport and Ortho–Para Fluctuation of Water Molecule in H2O@C60 Single Molecule Transistors, Nano Letters (2021). DOI: 10.1021/acs.nanolett.1c03604






