The 2017 Nobel Prize in chemistry has been awarded to Jacques Dubochet, formerly a professor at the University of Lausanne, Switzerland, Joachim Frank from the University of Columbia, and Royal Society Fellow Richard Henderson for their revolutionary work in bioimaging: the development of cryo-electron microscopy.

It’s really hard to get a good look at the stuff life is made of, known as ‘biomolecules’, in action. The issue is two-fold. For starters, they’re minuscule in size — we’re talking about chemical systems constructed out of bunches of atoms strung together. Secondly, the go-to tool for peering at the really small, electron beam imaging, just rips them to shreds before we can get an accurate picture.
The trio of scientists developed a technique to address these issues. Known as cryo-electron microscopy, it allows researchers to study the structure of biomolecules in high resolution without damaging them for the first time in history. The technology has immense potential in the field of biochemistry.
Frozen solid
Previously, electron microscopy imaging was only suitable for studying dead matter, because the electron beam destroys any biological material it is applied to. Henderson, a Scottish scientist and professor at the MRC Laboratory of Molecular Biology, was the first to use the method to generate three-dimensional images of a protein at the atomic scale. Joachim Frank, a professor at Colombia University in New York, expanded on electron microscopy, making it more flexible and more widely applicable. Together, their research made it easier to peer into the workings of biology’s building blocks in more detail than ever before.
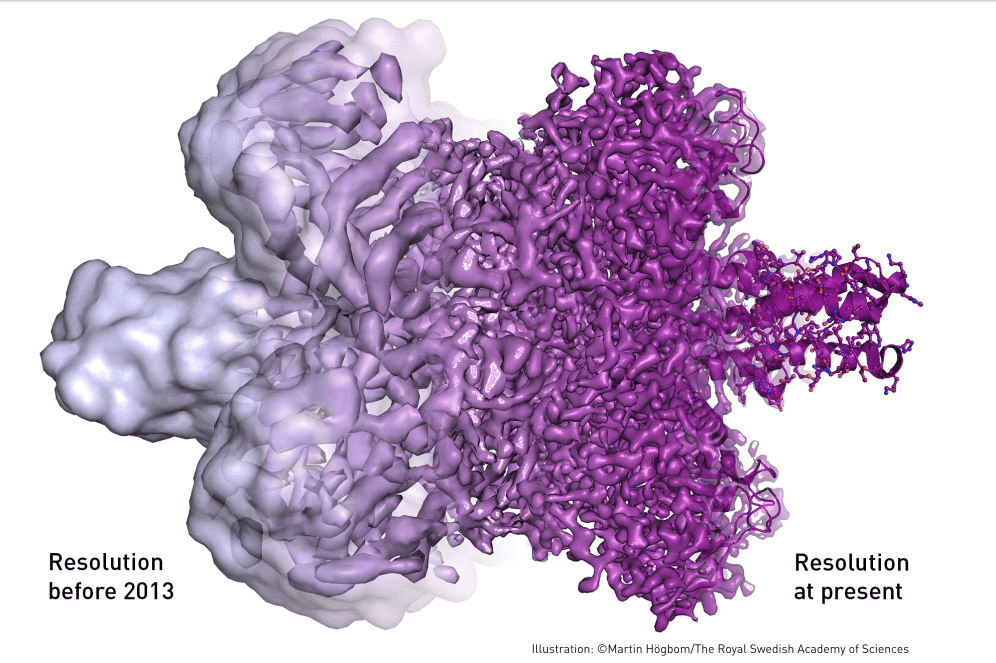
Image via NobelPrize.org
Dubochet, an honorary professor at the University of Lausanne in Switzerland, worked on making biomolecules stable enough to resist electron microscopy. His work refined a cold-vitrification technique, which made it possible to flash-freeze biomolecules without altering their original structures. This vitrification technique ensures that molecules can be kept intact (and still) while we take a good look at them using the advances in imaging achieved by Henderson and Frank. In a nutshell, that’s cryo-electron microscopy.
Practical applications of the technique are “immense”, Frank told journalists after the Prize’s announcement.
Cryo-electron microscopy is a game changer in biochemistry. Life at the smallest scale works much like a set of LEGO bricks. A virus trying to infect one of your cells, for example, first needs to bind its envelope proteins to those peppered throughout the cell’s membrane. Similarly, antibodies trying to hunt those viruses down bind to their envelope proteins to act as beacons, drawing in the immune system’s white cells. An antibiotic works by blocking proteins on the cells of bacteria, rendering them useless.
But not all types of proteins click together — they need to be ‘shaped’ properly. Up to now, scientists have had to simulate such structures from indirect observations. This took both a lot of time, processing power, or a lot of gamers putting in the elbow work. Now, researchers can simply use cryo-electron microscopy to look at the shapes they’re interested in.
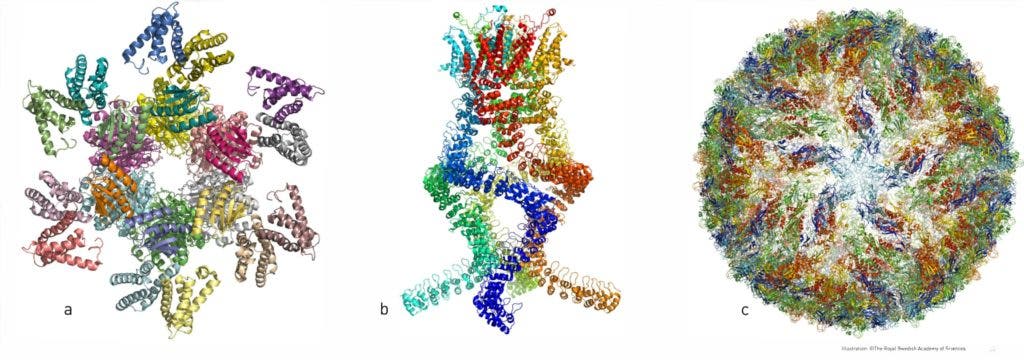
Image via NobelPrize.org
So far, it has allowed researchers to look at the structure of the Zika virus, the proteins that confer bacteria antibiotic resistance, and the enzyme producing the amyloid of Alzheimer’s disease. By freezing the same chemical systems at different points in its operation cycles, they can even put together film sequences of biochemistry at work — a feat unheard-of up to now.
Cryo-electron imaging, used in conjecture with a similar technique known as cryo-focused ion beam milling, was also applied by a team at ETH Zurich on some bigger pray — the bacterium Amoebophilus. Here, a cold-vitrification technique was used to strengthen the bacteria’s structures while keeping them brittle, and an ion beam was used to chip away bits of Amoebophilus. Finally, cryo-electron imaging was used to model the bacterium’s internal organelles, including a spectacular array of dagger-like projectile launchers.
All in all, this imaging technique promises to revolutionize our understanding of live biochemical processes, a much-needed aid in our growing antibiotic problem and beyond. In recognition of that fact, the Royal Swedish Academy of Sciences in Stockholm awarded the Nobel Prize, which will be equally shared among the three researchers on Wednesday.
Was this helpful?



