Researchers grew a miniature liver from human skin cells, and then they transplanted it into rats. The study could be a small step in developing a revolutionary technology that could help thousands of people who are waiting for a liver transplant.
This technology could help reduce organ shortage and it could also potentially lower the cost of a transplant. In the US alone, about 17,000 people are waiting for a liver transplant, according to Columbia University – a number that largely exceeds the amount available. At the same time, organ transplants are usually very expensive, especially in the US. In 2017, a patient receiving a liver transplant was billed over $800,000.
This development would also be very significant for the estimated 30 million people in the U.S. who have a liver disorder, according to the Health Resources and Services Administrator. The researchers are indeed optimistic.
“I believe it’s a very important step because we know it can be done,” co-author Alejandro Soto-Gutiérrez, a regenerative medicine researcher at the University of Pittsburgh, told Inverse. “You can make a whole organ that can be functional from one cell of the skin.”
Soto-Gutiérrez and his team of researchers collected skin cell samples from a group of human participants. Then they reprogrammed the skin cells into stem cells. After that, the cells were coaxed into various types of cells found in the human liver.
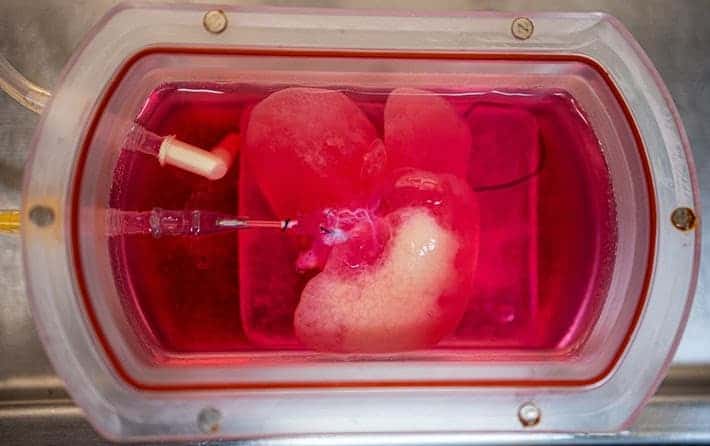
According to the paper, the scientists removed cells from rat livers so it could serve as a scaffolding for the stem cells they created. The human liver cells were seeded into this scaffolding, and finally, the livers were transplanted into the rats. It took under a month to grow the livers in bioreactors, while liver maturation usually takes up to two years.
Four days after the transplant, the researchers dissected the animals and discovered that the mini-livers had successfully worked. They saw that the rats had human liver proteins in their blood serum, and that with the mini-livers had secreted bile acids and urea just like a normal liver.
“Seeing that little human organ there inside the animal — brown, looking like a liver — that was pretty cool,” Soto-Gutiérrez told Inverse. “This thing that looks like a liver and functions like a liver came from somebody’s skin cells.”
Nevertheless, there were some unwanted side-effects. The researchers found problems with blood flow around the site of the graft (where the liver was transplanted). They argued that the following steps should address safety issues.
Soto-Gutiérrez and his team are working to create technology to enable widespread and access on-demand of human liver grafts that are functional.
“The long-term goal is to create organs that can replace organ donation, but in the near future, I see this as a bridge to transplant,” said Soto-Gutierrez. “For instance, in acute liver failure, you might just need a hepatic boost for a while instead of a whole new liver.”
The study was published in the journal Cell Reports.