
As electronics become ever thinner, smaller and faster, scientists always need to think ahead and develop solutions to accommodate the computing needs of the future. For one, it becomes clearer with each passing day that silicon – the most used material in electronics – can’t be used anymore for tomorrow’s tech since we’re nearing its maximum potential. Graphene, the wonder material consisting of carbon atoms arranged in a regular hexagon pattern, is considered by many silicon’s successor, ushering in a new age of electronics.
Graphene has been receiving a great deal of attention, and it’s no wonder why frankly. It’s enough to read some of the articles on ZME Science about it, or consider the fact that graphene research was awarded a $1 billion grant. Besides, its many uses, graphene has extraordinary electrical properties. Working with a material just one atom thick can be tricky, though, and developing working electronics with it on a mass scale (that’s the point after all) has proven to be a challenge.
Researchers at Stanford recently announced they successfully built smaller field-effect transistors (FETs) that use less power but operate faster, using ribbons of single-layer graphene laid side-by-side to create semiconductor circuits. What’s truly fantastic about their work is that they’ve created the graphene ribbons through a series of chemical reactions using DNA as an assembly mechanism.
DNA is ideal for assembling graphene since its roughly of the same dimensions. Chemically, DNA molecules contain carbon atoms, the material that forms graphene, so that’s another plus.
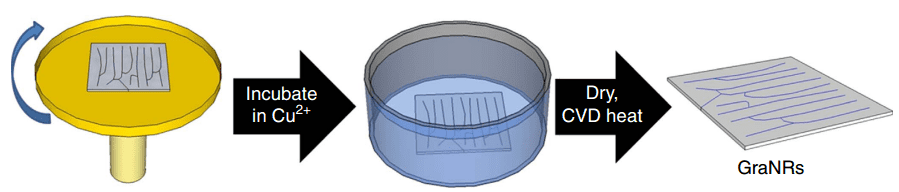
Stanford professor Zhenan Bao along with his team first started with a tiny platter of silicon to provide a support (substrate) for their experimental transistor. They dipped the silicon platter into a solution of DNA derived from bacteria and used a known technique to comb the DNA strands into relatively straight lines.
The DNA was then exposed to a copper salt solution, resulting in the copper ions being absorbed into the DNA. The platter was then bathed in methane gas (contains carbon atoms), and subjected to an increase in temperature until enough heat was generated for the sought after chemical reaction. The reaction freed some of the carbon atoms from the DNA and methane, which quickly joined together to form the infamous honeycomb graphene. Moreoever, since the separated carbon atoms more or less maintain their same position before breaking free, they formed ribbons that followed the structure of DNA.
Baking graphene with DNA
These ribbons are paramount to making working transistors that go into computer chips. Graphene in its typical structure can’t be used as a transistor, since it doesn’t have any bandgap (no electron state energy range) and thus can’t function as a semiconductor (switch on/off current). ZME Science reported a while ago how scientists at University of Riverside, California demonstrated that graphene transistors could work if one would exploit negative resistance.
The Stanford approach seems much simpler. Graphene, laterally confined within narrow ribbons less than 10 nanometers in width, exhibits a bandgap, meaning it can function as a semiconductor and thus solves graphene’s native issues.
The assembly process, two years in the making now, is far from being perfect. Not all carbon atoms formed honeycombed ribbons a single atom thick , and in some places the carbon atoms were displaced from their honeycombed structure altogether forming irregular patterns, leading the researchers to label the material graphitic instead of graphene.
“Our DNA-based fabrication method is highly scalable, offers high resolution and low manufacturing cost,” said co-author Fung Ling Yap. “All these advantages make the method very attractive for industrial adoption.”
Findings appeared in Nature Communications.
Was this helpful?



