Lifting the lockdown and moving on with our lives is predicated on massive testing for the coronavirus among the population. There’s really no way around it. An inexpensive new diagnostic test for COVID-19 based on the gene-editing tool CRISPR might prove extremely valuable in this regard. According to researchers at the UC San Francisco and Mammoth Biosciences who devised it, the test could offer a diagnosis in under 40 minutes.
Accurate testing at home by non-experts
The most commonly used COVID-19 test is known as RT-PCR, short for “reverse transcription-polymerase chain reaction”. It’s the same type of test that doctors use to diagnose HIV, measles, and mumps. This kind of test delivers an answer in 4-6 hours and requires expensive lab equipment and highly trained medical staff.
In contrast, the CRISPR test can return a result in well under an hour and can be performed at home, without the need for any special equipment — or the need for specially trained personnel. But how does it work?
CRISPR is a powerful gene-editing tool that allows scientists to dial in on specific bits of DNA inside cells and alter that piece of DNA. Since it was first unveiled in 2012, CRISPR technology has been used extensively to turn genes on and off without having to alter their sequence. But the technology has also proven highly controversial, especially after Chinese scientist He Jiankui used CRISPR on human embryos that ended up being carried to term and born in late 2018.
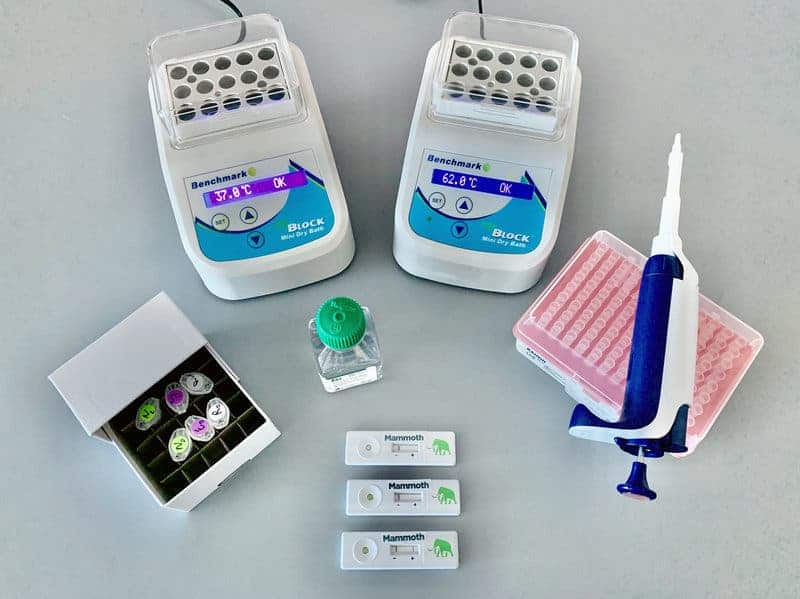
In this case, CRISPR in conjunction with a protein called Cas-12 and bits of viral genetic material designed to guide it, the researchers designed a test that targets two coronavirus genes (N and E genes).
Coronavirus tests designed by the CDC home in on the N gene while those made by the World Health Organization (WHO) target the E gene to spot COVID-19 cases. The new CRISPR test looks at both genes, allowing for a more robust (and quicker) diagnosis.
How it works
Like the standard PCR tests, the diagnosis first starts with a nasal swab from a patient. The swab is then introduced into the CRISPR-based test that can run multiple samples at once. When the CRISPR-Cas12 system recognizes genetic signatures from the coronavirus, a fluorescent molecule is released signaling the presence of the virus. The change of color determines whether a test is positive or not, similarly to a pregnancy test.
The test was developed in under three weeks and was trialed on a clinical sample of 36 COVID-19 patients and 42 patients with other viral respiratory infections. The results, which were published in the journal Nature Biotechnology, showed that the CRISPR-based DETECTR assay had a 95% positive predictive agreement and a 100% negative predictive agreement.
“The introduction and availability of CRISPR technology will accelerate deployment of the next generation of tests to diagnose COVID-19 infection,” said Charles Chiu, MD, PhD, professor of laboratory medicine at UCSF and co-lead developer of the new test
The process is much faster and less resource-intensive than traditional PCR-based tests. Although it is slightly less sensitive than PCR-based tests, the CRISPR kit can detect as few as 3.2 viral copies of the virus per microliter — a volume many times smaller than a drop of water. Since COVID-19 patients typically have a much higher viral load, the difference shouldn’t have a noticeable impact on diagnosis.
This test was developed in record time and there are many things that can be improved. Chiu and colleagues plan on turning the test into a handheld device with disposable cartridges that can be used by non-experts at home. The test is pending approval by the U.S. Food and Drug Administration (FDA) and once it passes regulatory roadblocks, it could soon enter mass production.






