In the beginning, the Earth’s surface was a lifeless, hot, but chemically rich place. In these harsh conditions, the first amino acids synthesized from inorganic compounds, and from them, proteins formed. They built the first single cells, which went on to form plants and animals. Recent research helps us understand the process that created amino acids, and there is a widespread consensus in the scientific community as to the path cells took to evolve to complex life as we know it today. But there is a missing link in the chain of events that ties lifeless to life: how and why did amino acids form the proteins that underpin the functions of every cell?
University of North Carolina scientists Richard Wolfenden and Charles Carter have shed new light on how the building blocks came together to form life some 4 billion years ago.
“Our work shows that the close linkage between the physical properties of amino acids, the genetic code, and protein folding was likely essential from the beginning, long before large, sophisticated molecules arrived on the scene,” said Carter, professor of biochemistry and biophysics at the UNC School of Medicine. “This close interaction was likely the key factor in the evolution from building blocks to organisms.”
Their findings, published in companion papers in the Proceedings of the National Academy of Sciences, contravene the traditional yet problematic “RNA world” theory, which posits that RNA -implicated in various biological roles in coding, decoding, regulation, and expression of genes- came into existence from the primordial pool, created the first simple proteins, peptides, and thus led to the creation of the first cells; in fact, they argue, it’s just as likely that peptides catalyzed the creation of RNA as the polymeric molecule was to lead to the formation of these simple proteins.
Image via: miltenyibiotec.com
The scientific community places the last universal common ancestor of all life on Earth, dubbed LUCA, at about 3.6 billion years ago. LUCA was most likely a single-cell organism, had a few hundred genes which stored the blueprints for DNA replication, protein synthesis, and RNA transcriptions. It had the basic components that modern organisms have, such at lipids. In other words, it had all the traits we expect to find in complex life today. After LUCA, it’s relatively easy to see how modern life evolved. But there is little hard evidence of how LUCA came into being.
We know a lot about LUCA and we are beginning to learn about the chemistry that produced building blocks like amino acids, but between the two there is a desert of knowledge,” Carter said. “We haven’t even known how to explore it.”
Dr. Wolfenden’s and Dr. Carter’s research aims to bridge the knowledge gap. It creates a model where RNA did not have to just pop into existance, and shows how even before there were cells, it seems more likely that there were interactions between amino acids and nucleotides that led to the co-creation of proteins and RNA.
“Dr. Wolfenden established physical properties of the twenty amino acids, and we have found a link between those properties and the genetic code,” Carter said. “That link suggests to us that there was a second, earlier code that made possible the peptide-RNA interactions necessary to launch a selection process that we can envision creating the first life on Earth.”
In order to function properly, proteins must fold in certain ways. The first PNAS paper, led by Wolfenden, shows that both the polarities of the amino acids (how they arrange themselves between water and oil) and their sizes help explain the complex process of protein folding – how a chain of connected amino acids arranges itself to form a particular 3-dimensional structure that has a specific biological function.
“Our experiments show how the polarities of amino acids change consistently across a wide range of temperatures in ways that would not disrupt the basic relationships between genetic coding and protein folding,” said Wolfenden, Alumni Distinguished Professor of Biochemistry and Biophysics.
This is an important piece of information, as when life started to form, in early-Earth conditions, temperatures were much hotter than when the first plants and animals appeared. If amino acid interaction changed with temperature in such a way as to interfere with genetic coding, the system would have stopped working when the planet cooled down. Life could not had evolved based on these principles.
A series of lab experiments with amino acids showed that two properties, namely size and polarity of amino acids, were necessary but also sufficient to explain how they behaved in folded proteins and that the relationship between the two held at the temperatures Earth had 4 billion years ago.
In their second PNAS paper, lead by Carter, they analyse how aminoacyl-tRNA synthetases recognized the enzymes that translate the genetic code, named transfer ribonucleic acid or tRNA. It shows that each one of the two ends of the L-shaped tRNA molecule has specific rules on what amino acid to tie to. The end that carried the amino-acid selects the molecule based on size. The other end, named an anticodon, selects it based on polarity:
“Think of tRNA as an adapter,” Carter said. “One end of the adapter carries a particular amino acid; the other end reads the genetic blueprint for that amino acid in messenger RNA. Each synthetase matches one of the twenty amino acids with its own adapter so that the genetic blueprint in messenger RNA faithfully makes the correct protein every time.”
Translating the genetic code is the nexus connecting pre-biotic chemistry to biology”, he added.
Their findings imply that the relationships between tRNA and the sizes and polarities of amino acids were crucial during the Earth’s early days. And basing on Carter’s work with the active cores of tRNA synthetases, called Urzymes, it seems likely that selection by size preceded selection after polarity. Because of this ordered selection, the earliest proteins did not have to fold into unique shapes, which evolved later. This would help explain two paradoxes: how complex structures arose from simple conditions, and how biology divided the work between proteins and nucleic acids, two very different structures.
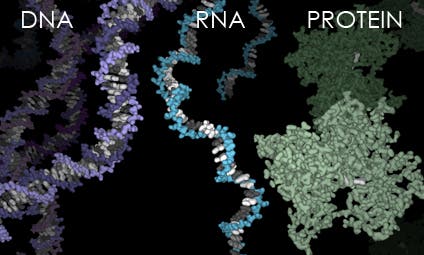
Image via: exploringorigins.org
“The fact that genetic coding developed in two successive stages — the first of which was relatively simple — may be one reason why life was able to emerge while the earth was still quite young,” Wolfenden noted.
“The collaboration between RNA and peptides was likely necessary for the spontaneous emergence of complexity,” Carter added. “In our view, it was a peptide-RNA world, not an RNA-only world.”
An earlier structure, that would enable coded peptides to bind RNA, would have provided a decisive selective advantage. This simple system would then undergo a natural selection process, paving the way for new and more biological forms of evolution.






