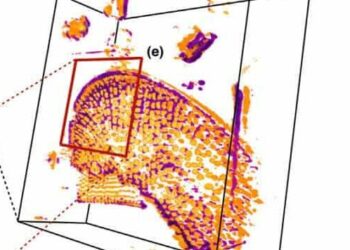
A new imaging technique called cryo-SR/EM can capture cells in unprecedented, 3D detail.
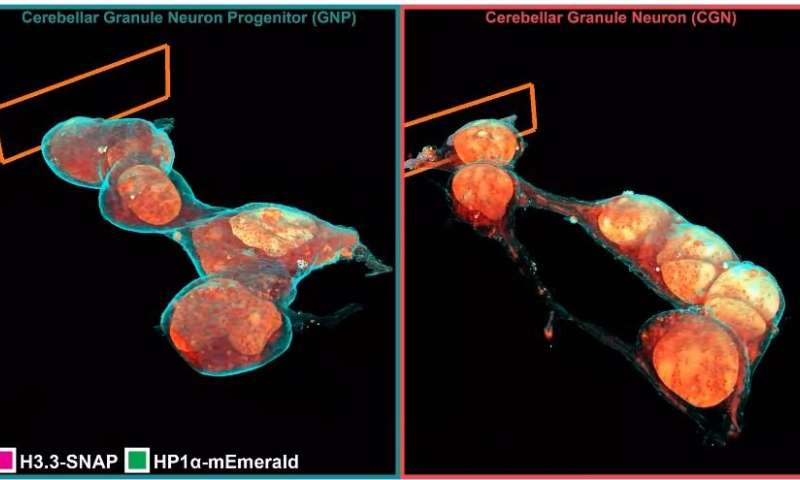
The insides of living cells are very cramped, busy, but fascinating places. Sadly, because they’re so small, we’ve never been able to take a proper look at what’s going on inside there.
However, a new technique that combines both optical and electron microscopy may allow us to do just that, and in very high detail to boot.
Zoomed in
“This is a very powerful method,” says Harald Hess, a senior group leader at the Howard Hughes Medical Institute’s Janelia Research Campus, US.
Cryo-SR/EM combines data captured from electron microscopy with high-resolution optical microscope imaging to create detailed 3D models of the inside of cells.
Separately, these two approaches are powerful but limited. Optical (or light) microscopes can easily differentiate between individual cell structures when fluorescent molecules are attached to them — this is known as super-resolution (SR) fluorescence microscopy. While it does provide a clear picture, SR fluorescence microscopy isn’t able to show all the proteins swishing about inside a cell at the same time, making it hard to see how different bits interact with everything else.
On the other hand, electron microscopy (EM) can ‘see’ virtually everything that’s happening inside a cell in very high detail, but it can be too powerful for its own sake. The wealth of features inside a cell, all seen in very high detail, can make it difficult to understand what you’re looking at.
The team worked to combine these two techniques into a single one that enhances their strengths while balancing out their respective limitations.
Cryo-SR/EM involves first freezing a cell or group of cells under high pressure; this step instantly freezes their internal activity without allowing for ice crystals to form (these can easily rupture cellular structures). Next, the samples are placed into a cryogenic chamber where they’re imaged in 3D using SR fluorescence microscopy. Finally, the samples are removed, embedded in resin, and viewed under an electron microscope (this paper used a powerful device developed in Hess’ lab). This step involves shooting a beam of ions at the cell, producing images of successively layers of the cell. A computer program is used to piece these images back together into a 3D reconstruction.
For the final step, all the data is pooled together, creating a very high-detail 3D model of the cell’s interior.
Techniques such as cryo-SR/EM promise to let us peek into natural systems that were previously just too tiny to spot — and they let us do so in extreme detail. One particularly-exciting possibility is that cryo-SR/EM will allow researchers to observe snapshots of cellular processes as they unfold, propelling forward basic science and its applications in fields such as genetic engineering, bioengineering, biochemistry, and medicine.
The paper “Correlative three-dimensional super-resolution and block-face electron microscopy of whole vitreously frozen cells” has been published in the journal Science.