Engineers working in the US have found a way to extract almost 100% of the water from brine, up from 6%. This innovation can alleviate water shortages in parts of the world where water is scarce, but it can also reduce the high salinity of disposable waste (ie in hydraulic fracking).
Water shortage is no joke. As the world population continues to grow, more and more areas use water unsustainably and are almost certainly set for a future water crisis. In these conditions, desalination becomes more and more a tempting option, as Israel has been demonstrating for a few years already. Still, the process can be significantly improved, as a team from University of California, Riverside, has proven.
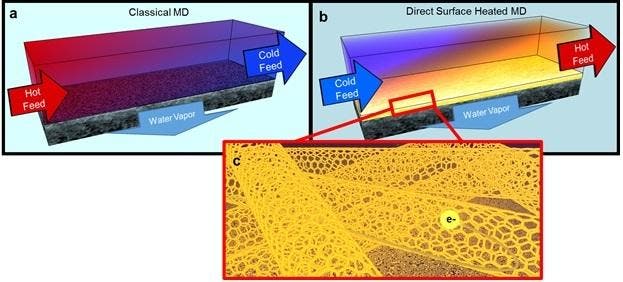
The team has developed a carbon nanotube heating element, vastly improving the recovery of fresh water during membrane distillation processes. Describing the new approach in the journal Nature Nanotechnology. David Jassby, an assistant professor of chemical and environmental engineering in UCR’s Bourns College of Engineering explains that previously, the recovery rate was capped at a much lower figure.
“In an ideal scenario, thermal desalination would allow the recovery of all the water from brine, leaving behind a tiny amount of a solid, crystalline salt that could be used or disposed of,” Jassby said. “Unfortunately, current membrane distillation processes rely on a constant feed of hot brine over the membrane, which limits water recovery across the membrane to about 6 percent.”
Most desalinization facilities use reverse osmosis, but the more salt you have in the water, the less efficient this process becomes. When dealing with brines, reverse osmosis becomes highly inefficient.
While such brines are rarely prevalent naturally, they are often produced as waste and must be disposed of to prevent environmental damage. What Jassby and his collaborators did not only ensure that all the water is desalinized, but it also reduces the necessary heat for the process, and thus saves a lot of energy.
The study has another interesting outcome — hot, briny water is a highly corrosive environment, and in order to develop this device, they also had to make sure that the parts can survive and operate properly for a longer period of time. Specifically, a threshold frequency was identified where electrochemical oxidation of the nanotubes was prevented.
Journal Reference: Alexander V. Dudchenko, Chuxiao Chen, Alexis Cardenas, Julianne Rolf & David Jassby — Frequency-dependent stability of CNT Joule heaters in ionizable media and desalination processes. doi:10.1038/nnano.2017.102






