New imaging techniques might revolutionize the technologies currently used to capture uranium from seawater, as researchers gain a better understanding of the way the compounds that bind the atoms interact with them.
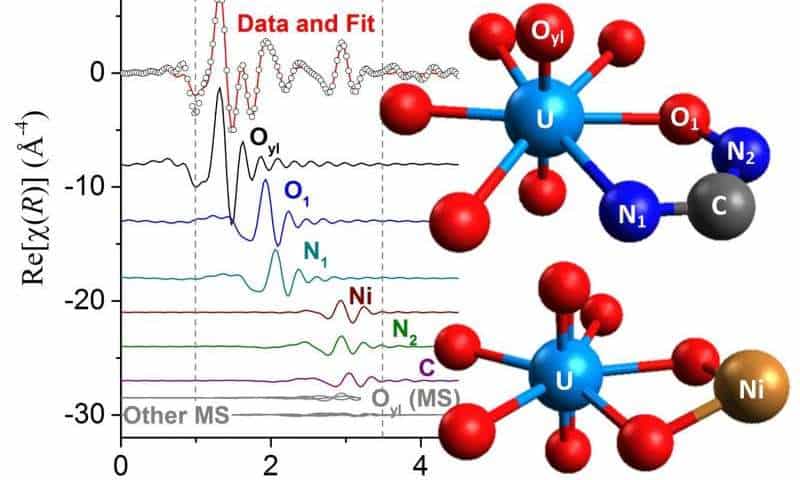
Image via phys
A research team led by Carter Abney, Wigner Fellow at the Department of Energy’s Oak Ridge National Laboratory, used ultra-high-resolution imaging to study the polymer fibers that bind uranium from seawater. Their results, gained through collaboration with the University of Chicago and published in a paper in the journal Energy & Environmental Science, shows that these materials don’t behave the way computational models say they should.
“Despite the low concentration of uranium and the presence of many other metals extracted from seawater, we were able to investigate the local atomic environment around uranium and better understand how it is bound by the polymer fibers,” Abney said.
By looking at the polymeric absorbent materials with X-ray Absorption Fine Structure spectroscopy at the Advanced Photon Source, Argonne National Laboratory, the researchers found that the spectrum response from the polymers were very different from what they were expecting to see based on previous small molecule and computational investigations.
They concluded that for this system the approach of studying small molecule structures and assuming that they accurately represent what happens in a bulk material simply doesn’t work. What is needed is to consider the behavior of the molecules in-bulk, to take into account interactions that only start working in a large-scale setting, says Abney.
“This challenges the long-held assumption regarding the validity of using simple molecular-scale approaches to determine how these complex adsorbents bind metals,” Abney said. “Rather than interacting with just one amidoxime, we determined multiple amidoximes would have to cooperate to bind each uranium molecule and that a second metal that isn’t uranium also participates in forming this binding site.”
(Amidoximes are the chemical group attached to the polymer fibers that bind the uranium atoms.)
Armed with this knowledge, Abney and colleagues hope to develop absorbents that can efficiently harvest the vast quantities of uranium dissolved in seawater.
“Nuclear power production is anticipated to increase with a growing global population, but estimates predict only 100 years of uranium reserves in terrestrial ores,” Abney said. “There is approximately 1,000 times that amount dissolved in the ocean, which would meet global demands for the foreseeable future.”






