For medication or vaccines to be okayed for use on humans, they first need to be extensively tested in the lab. Drugs need to be tested in living organisms, which is very different than in a petri dish because there are more complex interactions. Often, they are tested on mice, which are the prime model of human disease because they share 99% of our genes and are cheap and easy to test in the lab. However, sometimes, vaccine studies have wildly different results in mice, humans, and other animals. That could be due to the contents of mice guts, a new study reports. The lack of exposure to enough healthy bacteria may leave the immune system of lab mice compromised and not such a good model for human disease.

Natural is better
Laboratory mice have been bred to be genetically similar for reproducible results. In addition, the mice spend their entire lives in the lab and live mostly in sterilized environments. They are not exposed to the same bacteria that wild mice would be exposed to. These bacteria make up the microbiome, which is important in digestion and the immune system. As such, lab mice do not react to medication the same way that mice with a healthy community of gut bacteria would. In the same vein, humans don’t live in a sterilized box and have a more varied microbiome than lab mice and perhaps the reactions are not comparable.
Researchers from the National Institute of Diabetes and Digestive and Kidney Diseases tested how lab mice with a more natural microbiome would respond to diseases. They trapped closely related wild mice and sampled their bacterial gut microbiomes. Indeed, the wild mice had different gut biota than the laboratory mice. They took pregnant germ-free mice and they gave some of them a lab mouse microbiome and some a wild mouse microbiome and raised them over several generations. After four generations, the descendants had the same microbiomes as their ancestors were given.
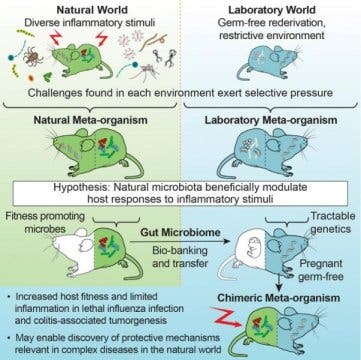
“We think that by restoring the natural ‘microbial identity’ of laboratory mice, we will improve the modeling of complex diseases of free-living mammals, which includes humans and their diseases,” said Barbara Rehermann, M.D., chief of the Immunology Section, Liver Diseases Branch, of the NIH’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK).
When exposed to the influenza virus, 92% of the mice with wild biomes survived while only 17% of control mice survived. They also had fewer tumors and less severe disease in the face of induced colorectal tumors. Reduced inflammation was observed in both cases. The wild microbiome could then be added to disease testing as an additional component. It could also explain why certain results in mice are not reproducible between labs, despite using the same strain of mice and other conditions. The microbiome may influence infection and drug response. This new model with “wild” gut bacteria seems to be more predictive for free-living mammals than current models. It shows the protective mechanisms that living creatures have that are relevant in the natural world.
Future outlook
Although not a goal of the study, it brings up another parallel between mice and men. The lab mice raised in a sterile environment were more vulnerable to disease and cancers. Children that are raised with few germs in the house have a higher chance of developing allergies and asthma. To some degree living in a germ-free or intensely disinfected house in addition to not spending much time outside lowers the number of healthy bacteria and keeps a healthy, diverse microbiome from forming. Perhaps now, or in the future, our ability to respond to diseases will diminish due to an unhealthy microbiome.
This study is not saying that all mouse-based studies are now rubbish. It is adding an extra tool to use in mouse-based research, in particular, for specific research questions.
“We do not intend to propose a general switch to natural microbiota in all types of studies. We rather think, that future research should aim to use microbiota tailored to specific research questions. For example, our findings are relevant in the context of the hygiene hypothesis, in that early exposure to inflammatory stimuli that are abundant in the natural world, has beneficial effects. We think that “natural microbiota” may be a valuable tool to further probe the hygiene hypothesis in regards to many unanswered questions (e.g. auto-immune diseases, autoimmunity, allergies etc..),” said Stephan Rosshart, M.D., first author of the paper and NIDDK postdoctoral fellow to ZMEScience.
Additionally, this new model is important for preclinical medical studies to try to better reflect real-world conditions. It could model human reactions to certain medications and treatment, and lead to new therapies. For example, changing the microbiome or imitating the same pathways may become a disease treatment. This study opens up many, interesting new directions to explore.
Journal reference: Rosshart et al. Wild Mouse Gut Microbiota Promotes Host Fitness and Improves Disease Resistance. Cell, 2017 DOI: 10.1016/j.cell.2017.09.016






