Researchers have produced E. coli bacteria with completely synthetic DNA. While the research was aimed at studying genetic redundancies, the potential applications are limitless.
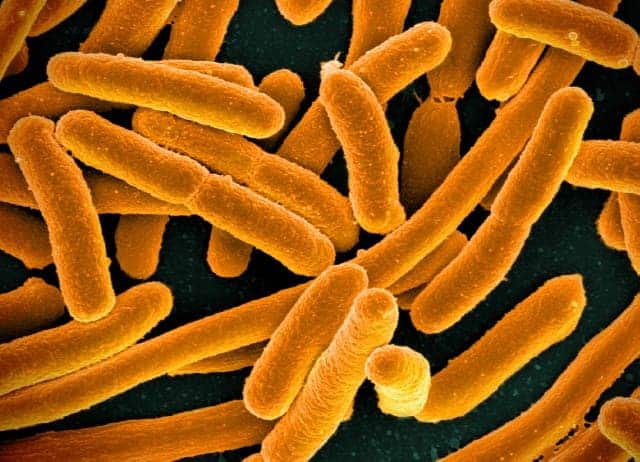
Researchers at the University of Cambridge have rewritten the DNA of the bacteria Escherichia coli, a strain of bacteria that is normally found in soil and the human gut. Although the bacteria look a bit weird and have some issues reproducing, they are alive and seem to function relatively normally — running by a set of rules directed by the human-edited genome.
For all its diversity and variation, all life on Earth (with the exception of some viruses) is based on DNA. The two-stranded DNA molecule is the blueprint for life as we know it, and each strand is composed of molecules containing just four bases: adenine, cytosine, guanine, and thymine (or A, C, G and T). Think of it this way: a handful of chemical letters are used to made three-letter words, and these words are then passed out as biological orders to proteins.
The four letters can be strung into 64 combinations of three-letter words called codons. Nearly all life on Earth uses these 64 codons, and these codons join to form virtually all proteins that can be found in nature — with the mention that three codons are used as punctuation marks, separating individual codons from one another.
However, there is a lot of redundancy within these combinations. Many combinations do the same thing, so they can theoretically be removed — but where do you stop? In order to study this, Jason Chin, an expert in synthetic biology who led the project, conducted a genetic “word swap”. He went through the bacteria’s DNA, and whenever he came across a particular codon (TCG, a codon that makes an amino acid called serine), he rewrote it as another one (AGC, which does the same job). He did this for three sets of codons, but the resulting genome was too long and complicated to brute-force in a cell — so instead, researchers split it into small segments and swapped them piece by piece inside the E. coli genome. By the time they were done, there were no natural segments in the bacteria’s DNA; the whole thing was synthetic.
The team then watched the bacteria go about its life. The first good news came immediately: it lived. It grew slower and presumably weaker than its “normal” version, but it was very much alive.
It’s not the first time a bacteria has been created with a synthetic DNA, but this achievement is by far the most complex achievement. In 2010, researchers from the J. Craig Venter Institute in Maryland created the first cell with synthetic DNA. In 2017, researchers at the Scripps Institute unveiled the first stable, semi-synthetic organism. Researchers are slowly starting to experiment with nature’s lifeforms and moving towards Life 2.0. Knowing which codons we need and which can be dropped is essential for this, especially as other groups are working on creating synthetic DNA for even more complex creatures such as baker’s yeast. The potential applications are limitless.
In addition to shedding new light on the chemical intricacies of DNA, this type of designer bacteria can also come in handy in the medical industry. They could, for instance, stop viral infections, or deliver diabetes or other compounds for treating serious conditions such as cancer and heart disease. After a certain point, you could even use it for more frivolous purposes, such as creating tastier bread or beer.
There is, however, a very strong impediment to this type of study: costs. Producing and inserting synthetic DNA is still an extremely expensive pursuit. Now that we know it can be done, we also know that in theory, you can recode anything. Actually having the know-how and the resources to do that remains a different matter.
The study has been published in Nature.



