Both children and adults with attention deficit hyperactivity disorder (ADHD) often have to take medication like Adderall and Ritalin to control their condition. But very soon, parents in the US will have access to a novel treatment for ADHD that doesn’t involve drugs or psychotherapy — it’s a medical device attached to a child’s forehead which zaps the nervous system with a mild electrical shock.
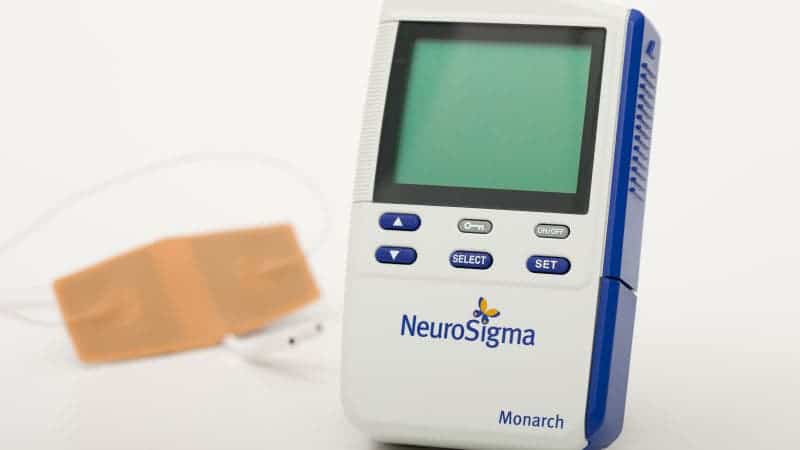
The device manufactured by NeuroSigma is called the Monarch external Trigeminal Nerve Stimulation (eTNS) System. Over the weekend, it was granted clearance by the Food and Drug Administration (FDA) for the treatment of children aged 7 to 12 who are currently not on any medication for their ADHD.
Attention-deficit hyperactivity disorder (ADHD) symptoms include inattention, hyperactivity, and acting impulsively. The CDC estimates that this disorder affects roughly 5% of under-18-year-olds (although the exact figure is likely higher) and two-thirds of those diagnosed continue to experience symptoms as adults.
NeuroSigma started designing their device in order to treat ADHD children who don’t respond to medication as well as to avoid the use of drugs that can have unpleasant side effects. Up to 25% of ADHD children do not respond to stimulants like Adderall or atomoxetine. The latter drug is typically the most effective but is also known to cause addiction, substance use disorder, and suicide ideation.
In the past decades, researchers have shown that stimulating certain parts of the brain with carefully tuned electric shocks can treat a range of neurological conditions like depression, epilepsy, and anxiety. Some approaches involve surgically implanting electrodes into the brain, however, the Monarch eTNS is far from being that extreme. The non-invasive, smartphone-sized device sends mild current to a patch attached on the forehead right above the eyebrows. The stimulation, which provokes a tingling sensation on the skin, makes its way through the cranial nerve to reach the cerebral cortex, which regulates emotions, behavior, and focus.
Earlier this year, researchers conducted a double-blinded, randomized, controlled trial involved 62 children with moderate to severe ADHD. One group was treated with the Monarch eTNS over a four-week period while a control group received a fake device that sent a sham electrical signal. At the end of the trial, those who were treated by the real device showed a significantly higher reduction in their ADHD symptoms than the control. The degree of improvement was on par with what you’d expect to see from non-stimulant treatment, such as behavioral therapy, which tends to be slightly less effective than drugs. However, the device makes up for this because of the lack of significant health risks — or none that we know of so far.
The device did produce some side effects such as drowsiness, trouble sleeping, teeth grinding, headaches, and fatigue, but these are quite mild compared to current pharmacological options. The price for the device will allegedly be equivalent to that of ADHD drugs, or just over $1,000. The same system can also be used to treat epilepsy and depression.






