
What happens when you mix the physical properties of glass (brittle and flowing) with those of metal (stiff and tough)? You get metal-glass, of course.
Since the 1960s, scientists showed you can make certain alloys into metal glass by rapidly cooling them. Really, really fast. Hundreds of degrees in a fraction of a second.
Eventually, you end up with an alloy that both behaves like a metal and glass. Some are three times stronger than titanium and have the elastic modulus of bone, all while being extremely lightweight. They’re also a lot easier to machine than pure metals.
All in all, metal glass is amazing and has the possibility to transform the world, just like another wonder material: graphene. So, why aren’t we seeing more of it?
Part of the problem is that research is moving painfully slow, but this may set to change after a team of researchers in Sydney reported a model for the atomic structure of metal glass. If until now scientists were testing various alloys and techniques in the dark, by trial and error, now they finally have a cookbook for metal glass.
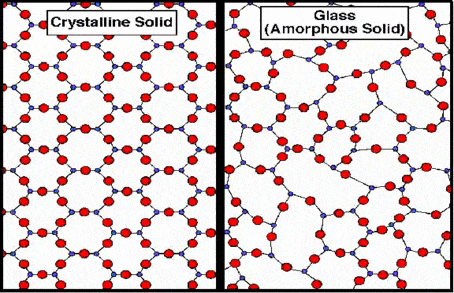
Glass might look like a solid, but in reality, it’s not exactly so. It’s both solid and liquid at the same time. It’s actually viscous, but it flows so slowly that it appears perfectly solid. Technically, glass is an amorphous solid, while metals are non-amorphous solids.
The difference between the two lies in the atomic structure. Glass, an amorphous solid, has its constituting atoms arranged in a chaotic, random pattern, while in metals there are arranged in an orderly crystalline lattice.
Reinforced glass isn’t exactly new. Corning’s Gorilla Glass coats cell phones, TVs, and other consumer electronics so it doesn’t instantly shatter when you drop it. Pyrex, used in telescope mirrors and baking dishes since 1915, is treated with heat to resist breakage.
But these sorts of glasses are only less brittle, not tough. Ideally, you want a type of glass you can use to build airplanes, cars, buildings, and even spaceships with. These sorts of applications require a material that is both tough and strong, which is why steel is so popular. But metal glass has a better combination of toughness and strength than any steel.
These amorphous glassy structures are metal alloys, composed of three or more metals such as magnesium, copper, and yttrium (Mg-Cu-Y). The mechanical and physical properties of BMGs depend strongly on the concentrations of the chemical elements that make up the alloy.
And when the correct combination of elements is heated or cooled at specific temperatures at specific rates the resulting materials can be extremely impressive. Some claim metal glass might revolutionize the world, second only to plastic in impact.
Identifying those alloys that can form metal glass is a tedious process, though. It has been estimated that only a minute fraction of the potential bulk metallic glass formers have been explored so far.
Researchers at the School of Materials Science and Engineering, New South Wales propose an alternate solution: fishing with a net instead of hooks. Specifically, they came up with a novel model of the atomic structure of metallic glass which allows them to predict which combination of metals will render the best metallic glasses.
No fewer than 200 new metallic glass alloys just based on titanium, magnesium, silver, copper, and zinc were showcased in the study published in Nature.
Now, it’s only a matter of shifting through each of these, systematically. The future sounds good.






