Researchers from a trio of U.S. universities have developed a technique to produce butadiene — a molecule traditionally sourced from oil or natural gas that underpins synthetic rubber and plastics — from renewable sources.

Image credits Hans Braxmeier.
Butadiene is the prime building block used for a whole bunch of materials we use today. It can be strewn together/polymerized to create styrene-butadiene rubber, the stuff quality tires are made of (apart, of course, from those made from eggshells and tomatoes). As nitrile butadiene rubber, it’s used to make hoses, seals, and the ubiquitous medical rubber glove. Butadiene is also the main component in acrylonitrile-butadiene-styrene, a rigid plastic that can be molded into hardy shapes — your computer or console case is likely made from this substance.
But getting your hands on butadiene does pose one economic and ecological problem — you need to refine natural hydrocarbons such as oil and gas to produce it. So understandably, there has been a push develop renewable (and if at all possible, cheaper) methods of obtaining this monomer. One new paper describes exactly one such method: the team — from the University of Delaware, the University of Minnesota and the University of Massachusetts — has invented a process to make butadiene from renewable sugars found in trees, grasses, and corn.
Our team’s success came from our philosophy that connects research in novel catalytic materials with a new approach to the chemistry,” says University of Delaware-based Catalysis Center for Energy Innovation Director Dionisios Vlachos, the Allan and Myra Ferguson Professor of Chemical and Biomolecular Engineering at UD and a co-author of the study. “This is a great example where the research team was greater than the sum of its parts.”
Our team combined a catalyst we recently discovered with new and exciting chemistry to find the first high-yield, low-cost method of manufacturing butadiene,” he adds. “This research could transform the multi-billion-dollar plastics and rubber industries.”
The three-step process begins with biomass-derived sugars. Using technology developed at the CCEI, the team can convert this sugars into a ring-like compound named farfural. This substance is then further processed into another ring compound called tetrahydrofuran (THF). The innovative third step uses phosphorus all-silica zeolite, a catalyst also developed at the CCEI, to break the THF rings into butadiene with more than 95 percent efficiency — considered a high-yield process in chemical manufacturing.
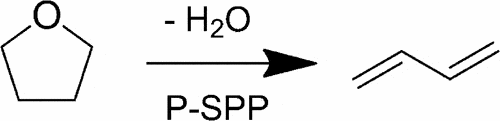
Image credits P. J. Dauenhauer et al., (2017), ACS.
The authors coined this novel, selective reaction “dehydra-decyclization” to show its capability for simultaneously removing water and cracking THF at once.
The paper “Biomass-Derived Butadiene by Dehydra-Decyclization of Tetrahydrofuran” has been published in the journal ACS Sustainable Chemistry & Engineering.






