Researchers at Brown University have found a cheaper and easier way to create hybrid perovskites, enabling engineers to develop more affordable and efficient solar cells.
Perovskite is a calcium titanium oxide mineral composed of calcium titanate (CaTiO3). The mineral has received much attention in recent years as artificial perovskite crystals have increasingly been used in solar cells. Perovskite films in solar cells are excellent light absorbers, but they until now, they were more expensive to fabric and only created small crystals.
“People have made good [perovskite] films over relatively small areas – a fraction of a centimeter or so square,” says researcher Nitin Padture, a professor of engineering.
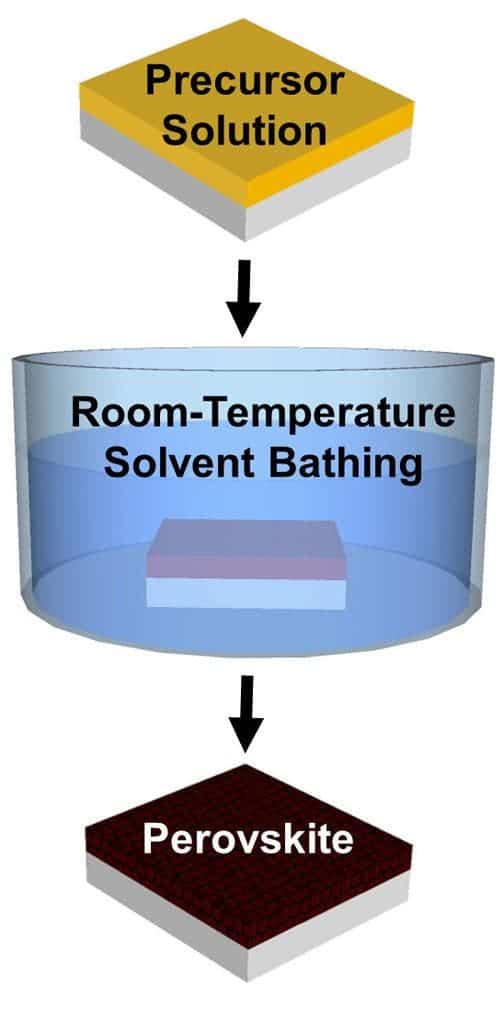
However, the research team developed a new technique, which only requires a room-temperature solvent bath to create perovskite crystals, rather than the blast of heat used in current crystallization methods.
The high temperature process involved temperatures of 100-150 degrees Celsius (212-302 Fahrenheit) and this also limited the kinds of substrates films can be deposited on. For example, plastics would make an excellent substrate, but you can’t really place these hot films on plastic because they’d just melt the plastic. The heat also created a tendency for crystals to form unevenly with tiny pinholes throughout the resulting film, which can reduce the efficiency of solar cells.
Yuanyuan Zhou, a graduate student in Padture’s lab, wanted to see if he could create perovskite crystals without having to use high temperatures and he came up with a solvent-solvent extraction method (SSE). He basically took the materials needed to create the crystals and dissolved them in a solvent called N-Methyl-2-pyrrolidone (NMP). Then, instead of heating, the substrate is bathed in a second solvent, diethyl ether (DEE). DEE selectively grabs the NMP and washes it away, and leaves behind a smooth film of perovskite crystals – exactly what researchers wanted to see. The entire process takes less than two minutes, it’s really cheap, and the crystals can be applied to any substrate. The SSE approach also ensures a very high quality and eliminates unwanted holes in the material.
“Using the other methods, when the thickness gets below 100 nanometers you can hardly make full coverage of film,” Zhou said. “You can make a film, but you get lots of pinholes. In our process, you can form the film evenly down to 20 nanometers because the crystallization at room temperature is much more balanced and occurs immediately over the whole film upon bathing.”
To make things even better, the new films are transparent, so we could be having transparent windows generating energy pretty soon. Zhou has also been able to make cells in different colors, to be used for decorative purposes. What I personally like a lot about this study is that unlike most solar cell developments which are strictly lab innovations and can take years to develop into something useful, this actually has the potential of becoming big soon.
“We think this could be a significant step toward a variety of commercially available perovskite cell products,” Padture said.
Journal Reference: Journal of Materials Chemistry A.
Was this helpful?



