
A crystal known to science for more than a century has only in recent years become recognized for its use in harvesting solar power. Since the first successful usage of perovskite in solar cells in 2009, the advances in the field have grown exponentially over time, making it a potential candidate for revamping the solar industry. Indeed, the crystal might just be what mainstream solar power has been waiting for: an easy to grow/manufacture solar harvesting platform that can be scaled.
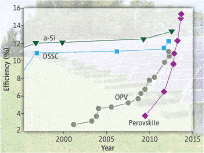
The perovskite mineral was originally found in the Ural Mountains in 1839, but it was only a few years ago that its ability to transport solar energy and convert it into electricity was discovered. Just a couple of years later, rated efficiency in the lab has soared from 3.8% to 19.3%, a pace of improvement unmatched by any other solar technology. Currently, the leading commercial solar tech employs crystalline silicon solar cells, which convert roughly 25% of incoming photon energy into electricity, but have decades worth of research backing them.
[RELATED] This material can be turned into a solar cell by day and light source by night
A promise, not a guarantee
What makes perovskite so interesting is its uncanny ability to diffuse photons a long distance through the cell when prepared in a liquid solution. Typically, solar cells convert energy to electricity by exploiting the hole-pair phenomenon. The photon hits the semiconducting material, then if its energy falls into the semiconductor bandcamp, an electron is offset, leaving a gap in the atom or hole. The electron travels from atom to atom within the material, occupying holes and offsetting at the same time until it eventually reaches an electrode and has its charge transferred to a circuit. Last step: profit and generate electricity.
The key is to have electron moving for as long as possible, and thanks to its diffusing capabilities, perovskite can theoretically generate more electricity. So far, however, what’s impressive about perovskite is how its been growing, not actual performance. Perovskite tech has seen 400 percent growth in solar conversion efficiency in less than five years. By contrast, traditional solar cells only increased their performance by 50 percent in their first five years.
It’s important to note that the upper end efficiencies come at a serious drawback – the complex crystalline structures are designed with lead. The resulting crystals readily dissolve in water or even humid air, which means people could be surprise to find dissolved lead is dripping from their rooftops. Not that great of a prospect, really. There’s a solution to this, however: replacing lead with tin, an element that sits right next to its toxic cousin in the periodic table and thus shares a similar electronic configuration.
Henry Snaith, a physicist at the University of Oxford in the United Kingdom, and colleagues report making tin perovskite solar cells that achieve a maximum efficiency of 6.4% – a very noticeable drop in efficiency, but there’s not reason to think this couldn’t be improved to match lead-based perovskite performance.
“There is no reason this new material can’t reach an efficiency better than 15 percent,” said Mecouri Kanatzidis, one of the researchers who published in Nature Photonics. “Tin and lead are in the same group in the periodic table, so we expect similar results.”
Tin perovskite is also easier to produce and integrate in current manufacturing processes. Also, the material can also be fine tuned to access different parts of the solar spectrum, so instead of using multiple semiconductors as is the case of >40% high-efficient multi-junction solar cells, you can use various tweaked layers made from the same crystal.
“It doesn’t require high-temperature processing. You can just dip glass into two chemicals and get the material to form on it,” said Joey Luther, a senior scientist at the Energy Department’s National Renewable Energy Laboratory (NREL).
According to N.R.E.L. senior scientist Joey Luther, the theoretical maximum efficiency of a perovskite-based solar cell would be around 31 percent.
“The goal shouldn’t be to stop at 20% efficiency,” Mr. Luther said. “The goal should be to try to get to 28% or higher. In the lab, the best cells need to be almost perfect at small scale. Then the commercial people can stop at whatever efficiency is economical for them to deploy.”
Clearly, perovskite momentum will definitely drop (a 400% growth in efficiency in the next five years is technically impossible), but it will remain to be seen just how far it can be integrated in commercial applications. Findings appeared in the journal Nature.






