They say that when two people fall in love, you can see sparks flying. Well, that may or may not be true, but researchers from the US have shown that when sperm meets and egg – sparks definitely fly.
Fertilization Fireworks
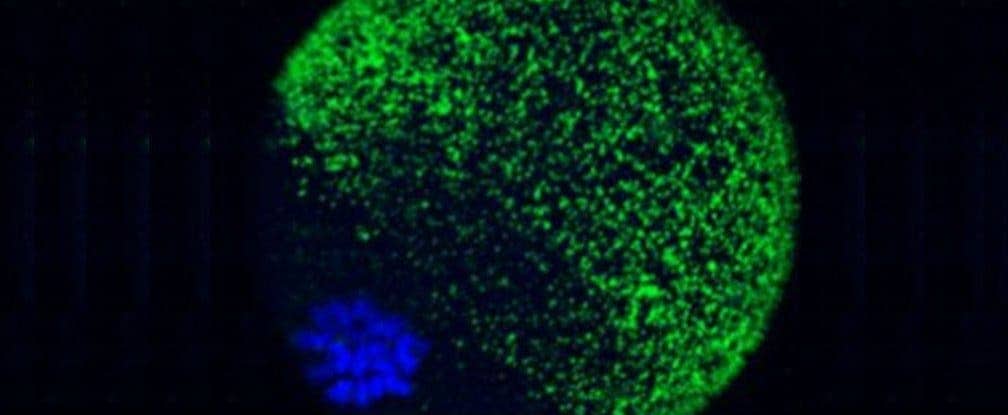
These are the first images captured at the exact moment when a mammal’s egg is fertilized, showing that in response, the egg releases billions of zinc atoms that create tiny ‘zinc sparks’ at the point of conception.
Northwestern University scientists had a hunch that this might be the case, so in order to prove their theory, they invented a a new fluorescent sensor that is able to track the movements of zinc in live cells. This allowed them to make a number of interesting findings, including an unprecedented view of an egg’s zinc-storage capabilities. The egg features some 8,000 zinc compartments, each one containing around one million zinc atoms; when it’s fertilized, it releases all those atoms in a firework-like display which lasts up to two hours.
The finding could lead to improved In Vitro Fertilization (IVF) results, showing which eggs have the highest quality.
“The amount of zinc released by an egg could be a great marker for identifying a high-quality fertilised egg, something we can’t do now,” said one of the team, ovarian biology expert, Teresa Woodruff, in the release. “If we can identify the best eggs, fewer embryos would need to be transferred during fertility treatments. Our findings will help move us toward this goal.”
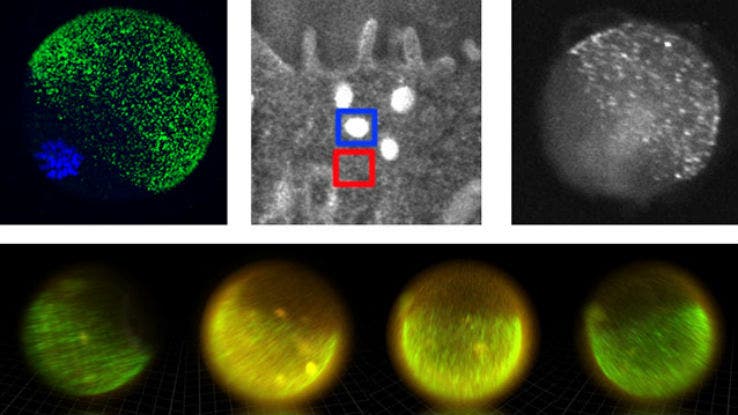
The team used high-energy X-Ray visualizing techniques to see where the zinc atoms are at any given moment.
“On cue, at the time of fertilisation, we see the egg release thousands of packages, each dumping a million zinc atoms, and then it’s quiet. Then there is another burst of zinc release,” said co-author, Thomas O’Halloran, in the release. “Each egg has four or five of these periodic sparks. It is beautiful to see, orchestrated much like a symphony. We knew zinc was released by the egg in huge amounts, but we had no idea how the egg did this.”
Why Zinc?
Of course, this begs the question – why does the egg eliminate so much zinc? Well, zinc has a lot to do with the egg’s decision to grow into an embryo. This is a very complex process, but as fertilization occurs and the egg “decides” it will grow into an embryo, it eliminates the zinc.
“The egg first has to stockpile zinc and then must release some of the zinc to successfully navigate maturation, fertilization and the start of embryogenesis,” said O’Halloran. “But exactly how much zinc is involved in this remarkable process and where is it in the cell? We needed data to better understand the molecular mechanisms at work as an egg becomes a new organism.”
The study, which was published in the journal Nature Chemistry, provides the first quantitative physical measurements of zinc localization in single cells in a mammal. The researchers are now working to see if they can somehow correlate zinc sparks to egg quality, but this study also lays the basis for understanding how zinc fluxes can regulate events in multiple biological systems beyond the egg, including neurotransmission from zinc-enriched neurons in the brain and insulin-release in the pancreas.
Journal Reference: Emily L. Que, Reiner Bleher, Francesca E. Duncan, Betty Y. Kong, Sophie C. Gleber, Stefan Vogt, Si Chen, Seth A. Garwin, Amanda R. Bayer, Vinayak P. Dravid, Teresa K. Woodruff & Thomas V. O’Halloran. Quantitative mapping of zinc fluxes in the mammalian egg reveals the origin of fertilization-induced zinc sparks. Nature Chemistrydoi:10.1038/nchem.2133
Was this helpful?