NASA scientists have noticed baffling seasonal changes in oxygen on Mars. The concentration of the gas, which many creatures on Earth require in order to breathe, rises and falls with the seasons in a way that scientists cannot yet explain, pointing towards mysterious chemical sources.

For the past six years that it has been on Mars, the Sample Analysis at Mars (SAM) mobile chemistry lab inside the Curiosity rover, has been sniffing the air above Gale Crater. The analysis confirmed the readings made by other science experiments since the 1970s, finding that the Martian atmosphere is made of 95% CO2, 2.6% nitrogen, 1.9% argon, 0.16% molecular oxygen (O2), and 0.06% carbon monoxide.
These molecules mix together and circulate around the planet due to changes in air pressure throughout the year. According to NASA, these seasonal changes are due to the freezing of CO2 over the poles during winter, which lowers the air pressure across the planet, and the evaporation of CO2 during spring and summer, which raises air pressure as the gas mixes across the Martian atmosphere.
The waxing and waning of CO2 concentrations at Gale Crater are followed by similar changes in nitrogen and argon — so, naturally, scientists thought that oxygen would follow the same curve. For some reason, though, this isn’t happening. Instead, the amount of oxygen in the air rises throughout spring and summer by as much as 30% and then drops back to predictable levels in fall. This pattern repeated each spring, however, the amount of oxygen added to the atmosphere varied — in other words, something must be producing it and something must be removing it.
“The first time we saw that, it was just mind-boggling,” said Sushil Atreya, professor of climate and space sciences at the University of Michigan in Ann Arbor.
What could explain this peculiar pattern? What could be adding oxygen to the atmosphere and what could be subtracting it?
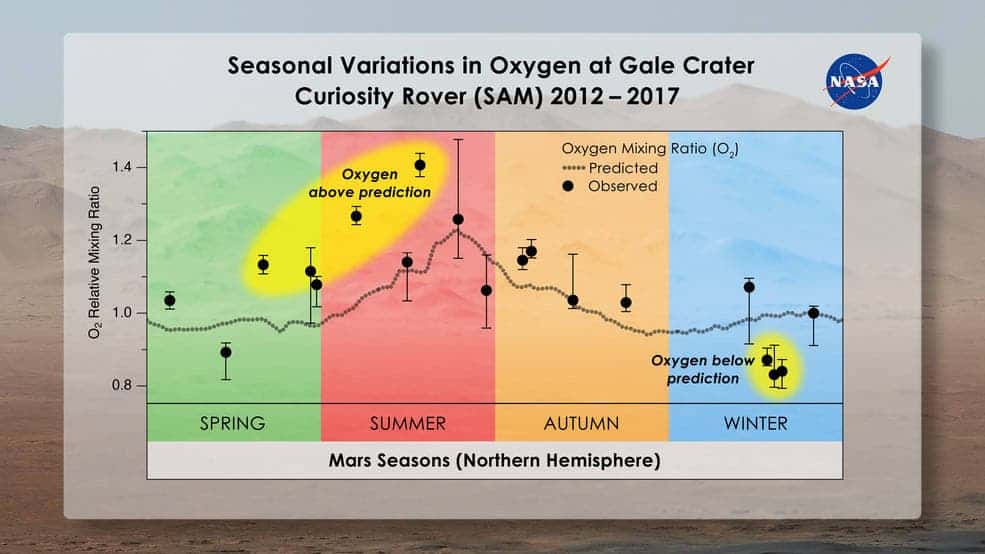
The SAM instrument itself is well calibrated and the readings are fine, NASA says. Perhaps, CO2 or water might have released the oxygen when the molecules were broken apart in the atmosphere. Later, solar radiation might break the molecular oxygen, leaving two single oxygen atoms free to escape into space. However, this explanation doesn’t stand because there would have to be five times more water than you can find on Mars to produce the extra oxygen and CO2 doesn’t break apart that fast. Moreover, it would take at least a decade for oxygen to break apart and disappear due to solar radiation.
There’s something out there that might explain this, but the truth is that, for now at least, scientists are left in the dark.
“We’re struggling to explain this,” said Melissa Trainer, a planetary scientist at NASA’s Goddard Space Flight Center in Greenbelt, Maryland who led this research. “The fact that the oxygen behavior isn’t perfectly repeatable every season makes us think that it’s not an issue that has to do with atmospheric dynamics. It has to be some chemical source and sink that we can’t yet account for.”
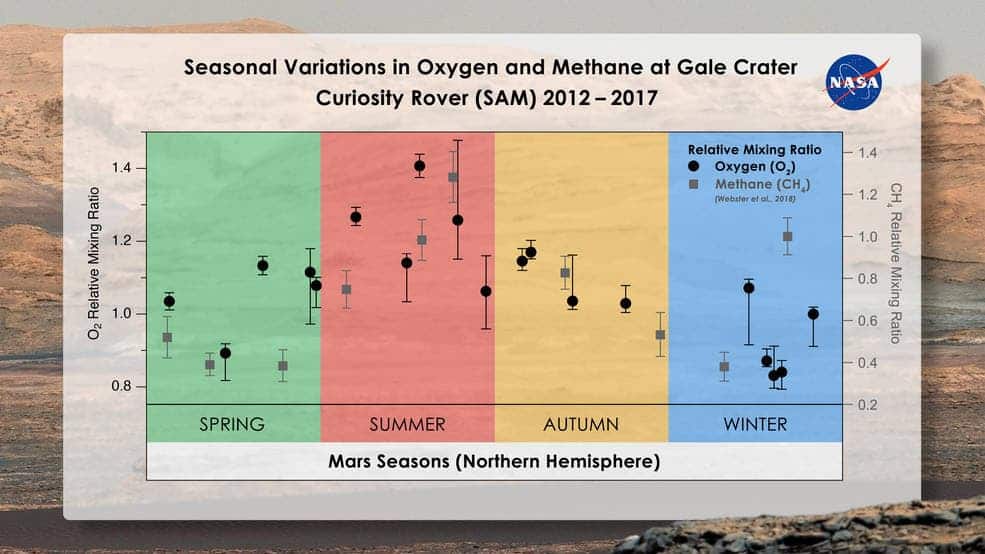
The explanation might be tied to another mysterious gas on Mars: methane. Since Curiosity arrived on Mars, the rover’s chemical sensors were able to detect methane, albeit in extremely minute quantities of 0.00000004% on average. The methane concentration also rises and falls seasonally, increasing by about 60% during the summer months. What’s more, the methane concentration in the atmosphere also spikes randomly and significantly at times. Again, scientists do not know why this is happening. But, what may be causing the spikes of methane could also be responsible for the skewed oxygen patterns. Sometimes, the two gases appear to fluctuate in tandem, for instance.
“We’re beginning to see this tantalizing correlation between methane and oxygen for a good part of the Mars year,” Atreya said. “I think there’s something to it. I just don’t have the answers yet. Nobody does.”
On Earth, oxygen and methane can both be produced by organisms but NASA says that on Mars their source isn’t likely to be biological. Instead, the gases are likely produced by chemical processes related to water and rock. One possible source for the extra springtime oxygen is the Martian soil, which contains hydrogen peroxide and perchlorates. Heat and humidity might release oxygen from the soil.
“We have not been able to come up with one process yet that produces the amount of oxygen we need, but we think it has to be something in the surface soil that changes seasonally because there aren’t enough available oxygen atoms in the atmosphere to create the behavior we see,” said Timothy McConnochie, assistant research scientist at the University of Maryland in College Park and another co-author of the paper.
The findings appeared in the Journal of Geophysical Research: Planets.



